Part 3: Adult Basic and Advanced Life Support
- On recognition of a cardiac arrest event, a layperson should simultaneously and promptly activate the emergency response system and initiate cardiopulmonary resuscitation (CPR).
- Performance of high-quality CPR includes adequate compression depth and rate while minimizing pauses in compressions.
- Early defibrillation with concurrent high-quality CPR is critical to survival when sudden cardiac arrest is caused by ventricular fibrillation or pulseless ventricular tachycardia.
- Administration of epinephrine with concurrent high-quality CPR improves survival, particularly in patients with nonshockable rhythms.
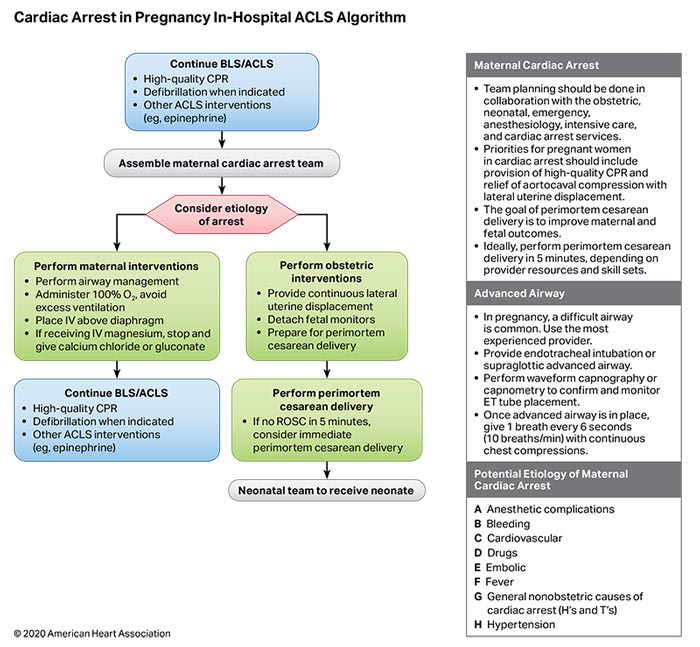
- Recognition that all cardiac arrest events are not identical is critical for optimal patient outcome, and specialized management is necessary for many conditions (eg, electrolyte abnormalities, pregnancy, after cardiac surgery).
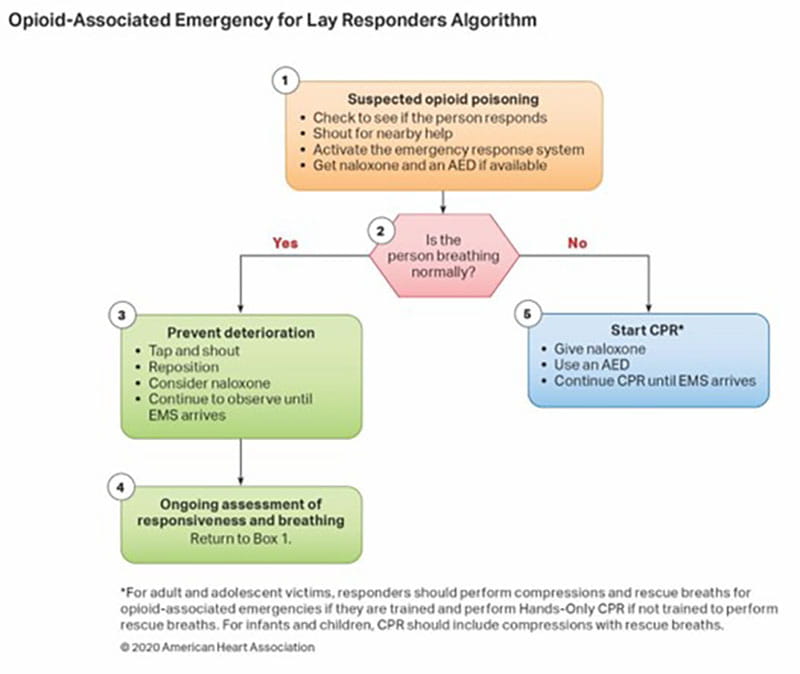
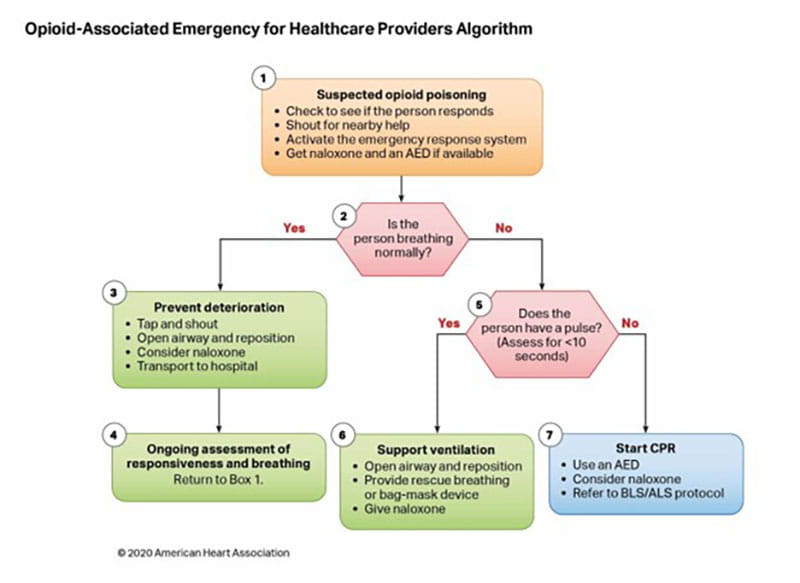
- The opioid epidemic has resulted in an increase in opioid-associated out-of-hospital cardiac arrest, with the mainstay of care remaining the activation of the emergency response systems and performance of high-quality CPR.
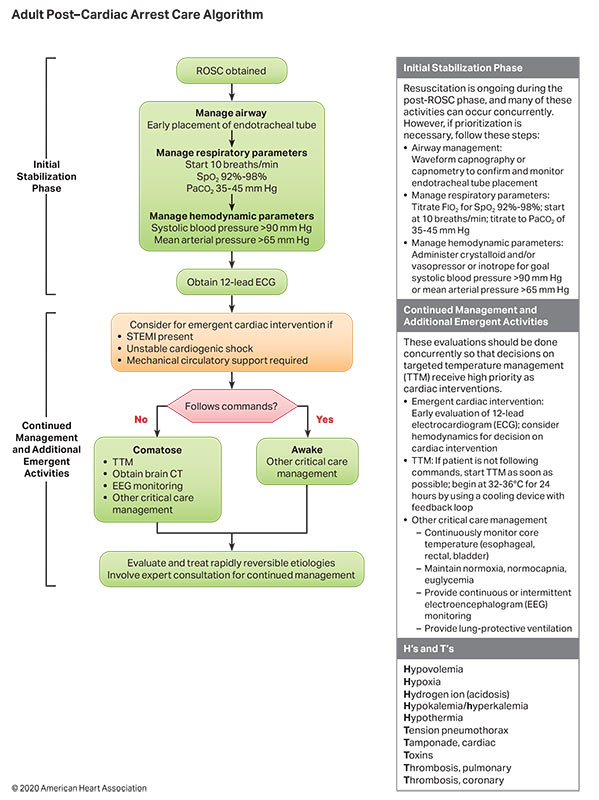
- Post–cardiac arrest care is a critical component of the Chain of Survival and demands a comprehensive, structured, multidisciplinary system that requires consistent implementation for optimal patient outcomes.
- Prompt initiation of targeted temperature management is necessary for all patients who do not follow commands after return of spontaneous circulation to ensure optimal functional and neurological outcome.
- Accurate neurological prognostication in brain-injured cardiac arrest survivors is critically important to ensure that patients with significant potential for recovery are not destined for certain poor outcomes due to care withdrawal.
- Recovery expectations and survivorship plans that address treatment, surveillance, and rehabilitation need to be provided to cardiac arrest survivors and their caregivers at hospital discharge to optimize transitions of care to home and to the outpatient setting.
In 2015, approximately 350 000 adults in the United States experienced nontraumatic out-of-hospital cardiac arrest (OHCA) attended by emergency medical services (EMS) personnel.1 Approximately 10.4% of patients with OHCA survive their initial hospitalization, and 8.2% survive with good functional status. The key drivers of successful resuscitation from OHCA are lay rescuer cardiopulmonary resuscitation (CPR) and public use of an automated external defibrillator (AED). Despite recent gains, only 39.2% of adults receive layperson-initiated CPR, and the general public applied an AED in only 11.9% of cases.1 Survival rates from OHCA vary dramatically between US regions and EMS agencies.2,3 After significant improvements, survival from OHCA has plateaued since 2012.
Approximately 1.2% of adults admitted to US hospitals suffer in-hospital cardiac arrest (IHCA).1 Of these patients, 25.8% were discharged from the hospital alive, and 82% of survivors have good functional status at the time of discharge. Despite steady improvement in the rate of survival from IHCA, much opportunity remains.
The International Liaison Committee on Resuscitation (ILCOR) Formula for Survival emphasizes 3 essential components for good resuscitation outcomes: guidelines based on sound resuscitation science, effective education of the lay public and resuscitation providers, and implementation of a well-functioning Chain of Survival.4
These guidelines contain recommendations for basic life support (BLS) and advanced life support (ALS) for adult patients and are based on the best available resuscitation science. The Chain of Survival, introduced in Major Concepts, is now expanded to emphasize the important component of survivorship during recovery from cardiac arrest, requires coordinated efforts from medical professionals in a variety of disciplines and, in the case of OHCA, from lay rescuers, emergency dispatchers, and first responders. In addition, specific recommendations about the training of resuscitation providers are provided in “Part 6: Resuscitation Education Science,” and recommendations about systems of care are provided in “Part 7: Systems of Care.”
These guidelines are designed primarily for North American healthcare providers who are looking for an up-to-date summary for BLS and ALS for adults as well as for those who are seeking more in-depth information on resuscitation science and gaps in current knowledge. The BLS care of adolescents follows adult guidelines. This Part of the 2020 American Heart Association (AHA) Guidelines for CPR and Emergency Cardiovascular Care includes recommendations for clinical care of adults with cardiac arrest, including those with life-threatening conditions in whom cardiac arrest is imminent, and after successful resuscitation from cardiac arrest.
Some recommendations are directly relevant to lay rescuers who may or may not have received CPR training and who have little or no access to resuscitation equipment. Other recommendations are relevant to persons with more advanced resuscitation training, functioning either with or without access to resuscitation drugs and devices, working either within or outside of a hospital. Some treatment recommendations involve medical care and decision-making after return of spontaneous circulation (ROSC) or when resuscitation has been unsuccessful. Importantly, recommendations are provided related to team debriefing and systematic feedback to increase future resuscitation success.
The Adult Cardiovascular Life Support Writing Group included a diverse group of experts with backgrounds in emergency medicine, critical care, cardiology, toxicology, neurology, EMS, education, research, and public health, along with content experts, AHA staff, and the AHA senior science editors. Each recommendation was developed and formally approved by the writing group.
The AHA has rigorous conflict of interest policies and procedures to minimize the risk of bias or improper influence during the development of guidelines. Before appointment, writing group members disclosed all commercial relationships and other potential (including intellectual) conflicts. These procedures are described more fully in “Part 2: Evidence Evaluation and Guidelines Development.” Disclosure information for writing group members is listed in Appendix 1(link opens in new window).
As with all AHA guidelines, each 2020 recommendation is assigned a Class of Recommendation (COR) based on the strength and consistency of the evidence, alternative treatment options, and the impact on patients and society (Table 1(link opens in new window)). The Level of Evidence (LOE) is based on the quality, quantity, relevance, and consistency of the available evidence. For each recommendation, the writing group discussed and approved specific recommendation wording and the COR and LOE assignments. In determining the COR, the writing group considered the LOE and other factors, including systems issues, economic factors, and ethical factors such as equity, acceptability, and feasibility. These evidence- review methods, including specific criteria used to determine COR and LOE, are described more fully in “Part 2: Evidence Evaluation and Guidelines Development.” The Adult Basic and Advanced Life Support Writing Group members had final authority over and formally approved these recommendations.
Unfortunately, despite improvements in the design and funding support for resuscitation research, the overall certainty of the evidence base for resuscitation science is low. Of the 250 recommendations in these guidelines, only 2 recommendations are supported by Level A evidence (high-quality evidence from more than 1 randomized controlled trial [RCT], or 1 or more RCT corroborated by high-quality registry studies.) Thirty-seven recommendations are supported by Level B-Randomized Evidence (moderate evidence from 1 or more RCTs) and 57 by Level B-Nonrandomized evidence. The majority of recommendations are based on Level C evidence, including those based on limited data (123 recommendations) and expert opinion (31 recommendations). Accordingly, the strength of recommendations is weaker than optimal: 78 Class 1 (strong) recommendations, 57 Class 2a (moderate) recommendations, and 89 Class 2b (weak) recommendations are included in these guidelines. In addition, 15 recommendations are designated Class 3: No Benefit, and 11 recommendations are Class 3: Harm. Clinical trials in resuscitation are sorely needed.
Open table in a new window.
| ACD | active compression-decompression |
| ACLS | advanced cardiovascular life support |
| ADC | apparent diffusion coefficient |
| AED | automated external defibrillator |
| AHA | American Heart Association |
| ALS | advanced life support |
| aOR | adjusted odds ratio |
| AV | atrioventricular |
| BLS | basic life support |
| COR | Class of Recommendation |
| CoSTR | International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations |
| CPR | cardiopulmonary resuscitation |
| CT | computed tomography |
| DWI | diffusion-weighted imaging |
| ECG | electrocardiogram |
| ECPR | extracorporeal cardiopulmonary resuscitation |
| EEG | electroencephalogram |
| EMS | emergency medical services |
| ETCO2 | (partial pressure of) end-tidal carbon dioxide |
| ETI | endotracheal intubation |
| GWR | gray-white ratio |
| ICU | intensive care unit |
| IHCA | in-hospital cardiac arrest |
| ILCOR | International Liaison Committee on Resuscitation |
| IO | intraosseous |
| ITD | impedance threshold device |
| IV | intravenous |
| LAST | local anesthetic systemic toxicity |
| LOE | Level of Evidence |
| MAP | mean arterial pressure |
| MRI | magnetic resonance imaging |
| NSE | neuron-specific enolase |
| OHCA | out-of-hospital cardiac arrest |
| Paco2 | arterial partial pressure of carbon dioxide |
| PCI | percutaneous coronary intervention |
| PE | pulmonary embolism |
| PMCD | perimortem cesarean delivery |
| pVT | pulseless ventricular tachycardia |
| RCT | randomized controlled trial |
| ROSC | return of spontaneous circulation |
| S100B | S100 calcium binding protein |
| SGA | supraglottic airway |
| SSEP | somatosensory evoked potential |
| STEMI | ST-segment elevation myocardial infarction |
| SVT | supraventricular tachycardia |
| TCA | tricyclic antidepressant |
| TOR | termination of resuscitation |
| TTM | targeted temperature management |
| VF | ventricular fibrillation |
| VT | ventricular tachycardia |
Survival and recovery from adult cardiac arrest depend on a complex system working together to secure the best outcome for the victim. The main focus in adult cardiac arrest events includes rapid recognition, prompt provision of CPR, defibrillation of malignant shockable rhythms, and post-ROSC supportive care and treatment of underlying causes. This approach recognizes that most sudden cardiac arrest in adults is of cardiac cause, particularly myocardial infarction and electric disturbances. Arrests without a primary cardiac origin (eg, from respiratory failure, toxic ingestion, pulmonary embolism [PE], or drowning) are also common, however, and in such cases, treatment for reversible underlying causes is important for the rescuer to consider.1 Some noncardiac etiologies may be particularly common in the in-hospital setting. Others, such as opioid overdose, are sharply on the rise in the out-of-hospital setting.2 For any cardiac arrest, rescuers are instructed to call for help, perform CPR to restore coronary and cerebral blood flow, and apply an AED to directly treat ventricular fibrillation (VF) or ventricular tachycardia (VT), if present. Although the majority of resuscitation success is achieved by provision of high-quality CPR and defibrillation, other specific treatments for likely underlying causes may be helpful in some cases.
The primary focus of cardiac arrest management for providers is the optimization of all critical steps required to improve outcomes. These include activation of the emergency response, provision of high-quality CPR and early defibrillation, ALS interventions, effective post-ROSC care including careful prognostication, and support during recovery and survivorship. All of these activities require organizational infrastructures to support the education, training, equipment, supplies, and communication that enable each survival. Thus, we recognize that each of these diverse aspects of care contributes to the ultimate functional survival of the cardiac arrest victim.
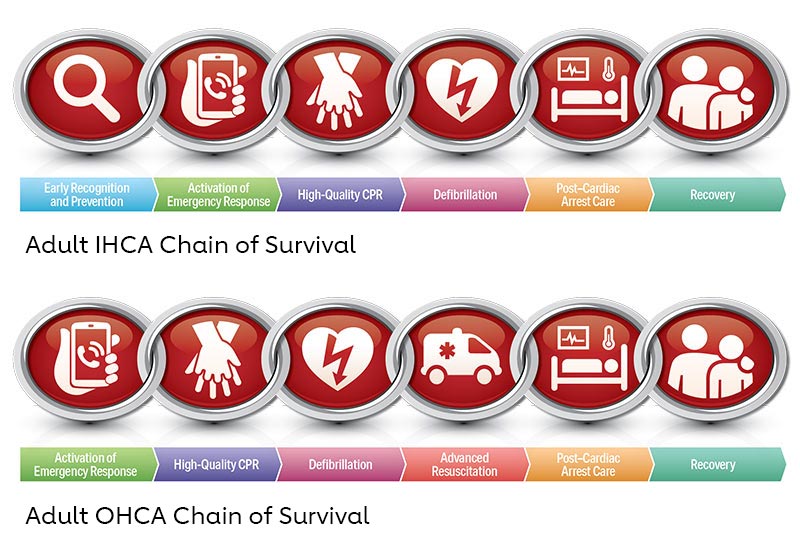
Resuscitation causes, processes, and outcomes are very different for OHCA and IHCA, which are reflected in their respective Chains of Survival (Figure 1). In OHCA, the care of the victim depends on community engagement and response. It is critical for community members to recognize cardiac arrest, phone 9-1-1 (or the local emergency response number), perform CPR (including, for untrained lay rescuers, compression-only CPR), and use an AED.3,4 Emergency medical personnel are then called to the scene, continue resuscitation, and transport the patient for stabilization and definitive management. In comparison, surveillance and prevention are critical aspects of IHCA. When an arrest occurs in the hospital, a strong multidisciplinary approach includes teams of medical professionals who respond, provide CPR, promptly defibrillate, begin ALS measures, and continue post-ROSC care. Outcomes from IHCA are overall superior to those from OHCA,5 likely because of reduced delays in initiation of effective resuscitation.
The Adult OHCA and IHCA Chains of Survival have been updated to better highlight the evolution of systems of care and the critical role of recovery and survivorship with the addition of a new link. This Recovery link highlights the enormous recovery and survivorship journey, from the end of acute treatment for critical illness through multimodal rehabilitation (both short- and long-term), for both survivors and families after cardiac arrest. This new link acknowledges the need for the system of care to support recovery, discuss expectations, and provide plans that address treatment, surveillance, and rehabilitation for cardiac arrest survivors and their caregivers as they transition care from the hospital to home and return to role and social function.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. If a victim is unconscious/unresponsive, with absent or abnormal breathing (ie, only gasping), the lay rescuer should assume the victim is in cardiac arrest. |
| 1 | C-LD | 2. If a victim is unconscious/unresponsive, with absent or abnormal breathing (ie, only gasping), the healthcare provider should check for a pulse for no more than 10 s and, if no definite pulse is felt, should assume the victim is in cardiac arrest. |
Synopsis
Lay rescuer CPR improves survival from cardiac arrest by 2- to 3-fold.1 The benefit of providing CPR to a patient in cardiac arrest outweighs any potential risk of providing chest compressions to someone who is unconscious but not in cardiac arrest. It has been shown that the risk of injury from CPR is low in these patients.2
It has been shown previously that all rescuers may have difficulty detecting a pulse, leading to delays in CPR, or in some cases CPR not being performed at all for patients in cardiac arrest.3 Recognition of cardiac arrest by lay rescuers, therefore, is determined on the basis of level of consciousness and the respiratory effort of the victim. Recognition of cardiac arrest by healthcare providers includes a pulse check, but the importance of not prolonging efforts to detect a pulse is emphasized.
Recommendation-Specific Supportive Text
- Agonal breathing is characterized by slow, irregular gasping respirations that are ineffective for ventilation. Agonal breathing is described by lay rescuers with a variety of terms including, abnormal breathing, snoring respirations, and gasping.4 Agonal breathing is common, reported as being present in up to 40% to 60% of victims of OHCA.5 The presence of agonal breathing is cited as a common reason for lay rescuers to misdiagnose a patient as not being in cardiac arrest.6 In patients who are unresponsive, with absent or abnormal breathing, lay rescuers should assume the patient is in cardiac arrest, call for help, and promptly initiate CPR. These 2 criteria (patient responsiveness and assessment of breathing) have been shown to rapidly identify a significant proportion of patients who are in cardiac arrest, allowing for immediate initiation of lay rescuer CPR. Further, initiation of chest compressions in patients who are unconscious but not in cardiac arrest is associated with low rates of significant adverse events.2 The adverse events noted included pain in the area of chest compressions (8.7%), bone fracture (ribs and clavicle) (1.7%), and rhabdomyolysis (0.3%), with no visceral injuries described.2
- Protracted delays in CPR can occur when checking for a pulse at the outset of resuscitation efforts as well as between successive cycles of CPR. Healthcare providers often take too long to check for a pulse7,8 and have difficulty determining if a pulse is present or absent.7–9 There is no evidence, however, that checking for breathing, coughing, or movement is superior to a pulse check for detection of circulation.10 Thus, healthcare providers are directed to quickly check for a pulse and to promptly start compressions when a pulse is not definitively palpated.9,11
This topic last received formal evidence review in 2010.3
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. All lay rescuers should, at minimum, provide chest compressions for victims of cardiac arrest. |
| 1 | C-LD | 2. After identifying a cardiac arrest, a lone responder should activate the emergency response system first and immediately begin CPR. |
| 1 | C-LD | 3. We recommend that laypersons initiate CPR for presumed cardiac arrest, because the risk of harm to the patient is low if the patient is not in cardiac arrest. |
| 2a | C-LD | 4. For lay rescuers trained in CPR using chest compressions and ventilation (rescue breaths), it is reasonable to provide ventilation (rescue breaths) in addition to chest compressions for the adult in OHCA |
Synopsis
After cardiac arrest is recognized, the Chain of Survival continues with activation of the emergency response system and initiation of CPR. The prompt initiation of CPR is perhaps the most important intervention to improve survival and neurological outcomes. Ideally, activation of the emergency response system and initiation of CPR occur simultaneously. In the current era of widespread mobile device usage and accessibility, a lone responder can activate the emergency response system simultaneously with starting CPR by dialing for help, placing the phone on speaker mode to continue communication, and immediately commencing CPR. In the rare situation when a lone rescuer must leave the victim to dial EMS, the priority should be on prompt EMS activation followed by immediate return to the victim to initiate CPR.
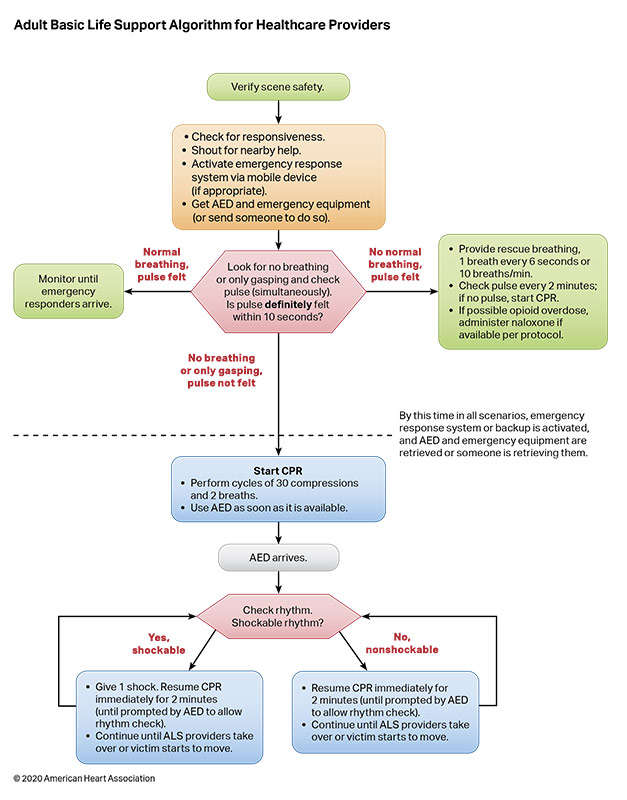
Existing evidence suggests that the potential harm from CPR in a patient who has been incorrectly identified as having cardiac arrest is low.1 Overall, the benefits of initiation of CPR in cardiac arrest outweigh the relatively low risk of injury for patients not in cardiac arrest. The initial phases of resuscitation once cardiac arrest is recognized are similar between lay responders and healthcare providers, with early CPR representing the priority. Lay rescuers may provide chest compression– only CPR to simplify the process and encourage CPR initiation, whereas healthcare providers may provide chest compressions and ventilation (Figures 2–4).
Recommendation-Specific Supportive Text
- CPR is the single-most important intervention for a patient in cardiac arrest, and chest compressions should be provided promptly. Chest compressions are the most critical component of CPR, and a chest compression–only approach is appropriate if lay rescuers are untrained or unwilling to provide respirations. Beginning the CPR sequence with compression.2–4 Nationwide dissemination of chest compression–only CPR for lay rescuers was associated with an increase in the incidence of survival with favorable neurological outcome after OHCAs in Japan, likely due to an increase in lay rescuers providing CPR.5 Chest compressions should be provided as soon as possible, without the need to remove the victim’s clothing first.
- The optimal timing of CPR initiation and emergency response system activation was evaluated by an ILCOR systematic review in 2020.1 An observational study of over 17 000 OHCA events reported similar results from either a “call-first” strategy or a “CPR-first” strategy.6 In the current era of ubiquitous mobile devices, ideally both the call to activate EMS and the initiation of CPR can occur simultaneously.
- Four observational studies7–10 reported outcomes from patients who were not in cardiac arrest and received CPR by lay rescuers. No serious harm from CPR was found in patients when they were later determined not to have been in cardiac arrest.1 This is in contrast to the significant risk of withholding CPR when a patient is in cardiac arrest, making the risk:benefit ratio strongly in favor of providing CPR for presumed cardiac arrest.
- In some observational studies, improved outcomes have been noted in victims of cardiac arrest who received conventional CPR (compressions and ventilation) compared with those who received chest compressions only.5,11,12 Other studies have reported no difference in outcomes for patients receiving conventional versus compression-only CPR.11,13–21 Given the potential benefit of conventional CPR, if lay rescuers are appropriately trained, they should be encouraged to concurrently deliver ventilation with compressions. A thorough review of the data concerning the ratio of compressions to ventilation when performing conventional CPR is discussed in Ventilation and Compression-to-Ventilation Ratio.
These recommendations are supported by the 2020 ILCOR Consensus on CPR and Emergency Cardiovascular Care Science With Treatment Recommendations (CoSTR).1
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. A lone healthcare provider should commence with chest compressions rather than with ventilation. |
| 2a | C-LD | 2. It is reasonable for healthcare providers to perform chest compressions and ventilation for all adult patients in cardiac arrest from either a cardiac or noncardiac cause. |
Recommendation-Specific Supportive Text
- The 2010 Guidelines for CPR and Emergency Cardiovascular Care included a major change for trained rescuers, who were instructed to begin the CPR sequence with chest compressions rather than with breaths (circulation, airway, and breathing versus airway, breathing, and circulation) to minimize the time to initiation of chest compressions. This approach is resupported by new literature, summarized in a 2020 ILCOR systematic review (Table 2).1–4 In the recommended sequence, once chest compressions have been started, a single trained rescuer delivers rescue breaths by mouth to mask or by bag-mask device to provide oxygenation and ventilation. Manikin studies demonstrate that starting with chest compressions rather than with ventilation is associated with faster times to chest compressions,3,23 rescue breaths,4 and completion of the first CPR cycle.4
- Healthcare providers are trained to deliver both compressions and ventilation. Delivery of chest compressions without assisted ventilation for prolonged periods could be less effective than conventional CPR (compressions plus ventilation) because arterial oxygen content decreases as CPR duration increases. This concern is especially pertinent in the setting of asphyxial cardiac arrest.11 Healthcare providers, with their training and understanding, can realistically tailor the sequence of subsequent rescue actions to the most likely cause of arrest.
These recommendations are supported by the 2020 CoSTR for BLS.1
Table 2. Adult BLS Sequence22
| Step | Lay Rescuer Not Trained | Lay Rescuer Trained | Healthcare Provider |
|---|---|---|---|
| 1 | Ensure scene safety. | Ensure scene safety. | Ensure scene safety. |
| 2 | Check for response. | Check for response. | Check for response. |
| 3 | Shout for nearby help. Phone or ask someone to phone 9-1-1 (the phone or caller with the phone remains at the victim's side, with the phone on speaker mode). | Shout for nearby help and activate the emergency response system (9-1-1, emergency response). If someone responds, ensure that the phone is at the side of the victim if at all possible. | Shout for nearby help/activate the resuscitation team; the provider can activate the resuscitation team at this time or after checking for breathing and pulse. |
| 4 | Follow the telecommunicator’s* instructions. | Check for no breathing or only gasping; if none, begin CPR with compressions. | Check for no breathing or only gasping and check pulse (ideally simultaneously). Activation and retrieval of the AED/emergency equipment by the lone healthcare provider or by the second person sent by the rescuer must occur no later than immediately after the check for no normal breathing and no pulse identifies cardiac arrest. |
| 5 | Look for no breathing or only gasping, at the direction of the telecommunicator. | Answer the dispatcher’s questions, and follow the telecommunicator’s instructions. | Immediately begin CPR, and use the AED/ defibrillator when available |
| 6 | Follow the telecommunicator’s instructions. | Send the second person to retrieve an AED, if one is available. | When the second rescuer arrives, provide 2-rescuer CPR and use the AED/defibrillator. |
AED indicates automated external defibrillator; BLS, basic life support; and CPR, cardiopulmonary resuscitation.
*Telecommunicator and dispatcher are terms often used interchangeably.
Introduction
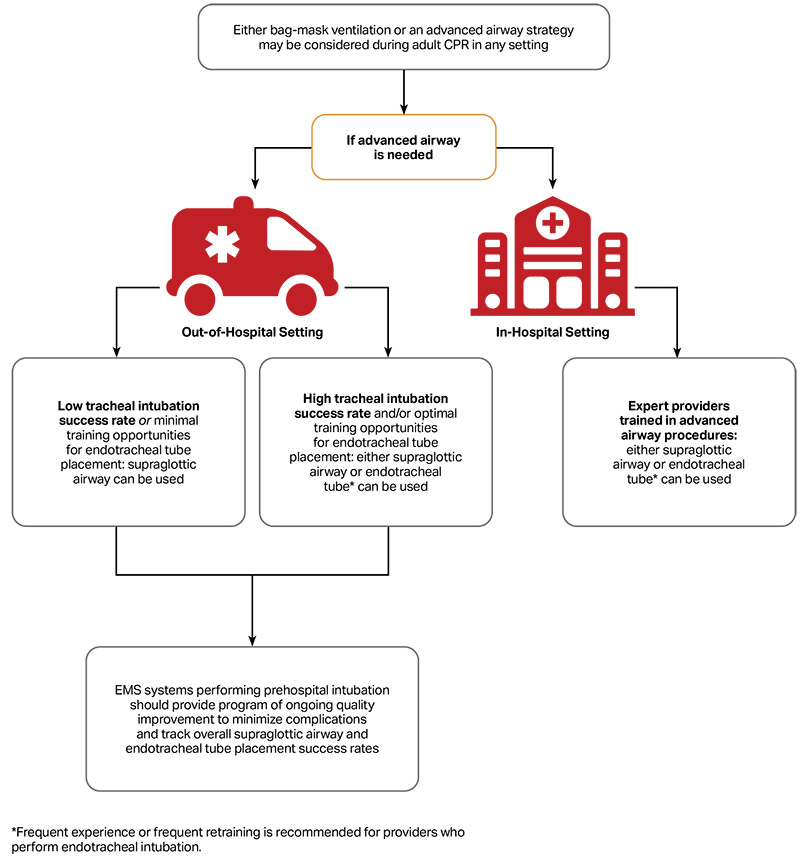
A patent airway is essential to facilitate proper ventilation and oxygenation. Although there is no high-quality evidence favoring one technique over another for establishment and maintenance of a patient’s airway, rescuers should be aware of the advantages and disadvantages and maintain proficiency in the skills required for each technique. Rescuers should recognize that multiple approaches may be required to establish an adequate airway. Patients should be monitored constantly to verify airway patency and adequate ventilation and oxygenation. There are no studies comparing different strategies of opening the airway in cardiac arrest patients. Much of the evidence examining the effectiveness of airway strategies comes from radiographic and cadaver studies.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-EO | 1. A healthcare provider should use the head tilt–chin lift maneuver to open the airway of a patient when no cervical spine injury is suspected. |
| 1 | C-EO | 2. The trained lay rescuer who feels confident in performing both compressions and ventilation should open the airway using a head tilt–chin lift maneuver when no cervical spine injury is suspected. |
| 2b | C-EO | 3. The use of an airway adjunct (eg, oropharyngeal and/or nasopharyngeal airway) may be reasonable in unconscious (unresponsive) patients with no cough or gag reflex to facilitate delivery of ventilation with a bag-mask device. |
| 2a | C-EO | 4. In the presence of known or suspected basal skull fracture or severe coagulopathy, an oral airway is preferred compared with a nasopharyngeal airway. |
| 3: No Benefit | C-LD | 5. The routine use of cricoid pressure in adult cardiac arrest is not recommended |
Recommendation-Specific Supportive Text
- and 2. The head tilt–chin lift has been shown to be effective in establishing an airway in noncardiac arrest and radiological studies.2–5 No studies have compared head tilt–chin lift with other airway maneuvers to establish an airway during cardiac arrest.
- Although there is no evidence examining the effectiveness of their use during cardiac arrest, oropharyngeal and nasopharyngeal airways can be used to maintain a patent airway and facilitate appropriate ventilation by preventing the tongue from occluding the airway. Incorrect placement, however, can cause an airway obstruction by displacing the tongue to the back of the oropharynx.6,7
- The benefit of an oropharyngeal compared with a nasopharyngeal airway in the presence of a known or suspected basilar skull fracture or severe coagulopathy has not been assessed in clinical trials. However, an oral airway is preferred because of the risk of trauma with a nasopharyngeal airway. Multiple case reports have observed intracranial placement of nasopharyngeal airways in patients with basilar skull fractures.8,9
- There is no evidence that cricoid pressure facilitates ventilation or reduces the risk of aspiration in cardiac arrest patients. There is some evidence that in non–cardiac arrest patients, cricoid pressure may protect against aspiration and gastric insufflation during bag-mask ventilation.10–13 However, cricoid pressure may also impede ventilation and the placement of a supraglottic airway (SGA) or intubation,14–20 and increase the risk of airway trauma during intubation.21
This topic last received formal evidence review in 2010.22
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-EO | 1. In cases of suspected cervical spine injury, healthcare providers should open the airway by using a jaw thrust without head extension. |
| 1 | C-EO | 2. In the setting of head and neck trauma, a head tilt–chin lift maneuver should be performed if the airway cannot be opened with a jaw thrust and airway adjunct insertion. |
| 3: Harm | C-LD | 3. In the setting of head and neck trauma, lay rescuers should not use immobilization devices because their use by untrained rescuers may be harmful. |
Recommendation-Specific Supportive Text
- Healthcare providers should consider the possibility of a spinal injury before opening the airway. If a spinal injury is suspected or cannot be ruled out, providers should open the airway by using a jaw thrust instead of head tilt–chin lift.2
- Maintaining a patent airway and providing adequate ventilation and oxygenation are priorities during CPR. If a jaw thrust and/or insertion of an airway adjunct are ineffective in opening the airway and allowing ventilation to occur, a head tilt–chin lift may be the only way to open the airway. In these cases, this maneuver should be used even in cases of potential spinal injury because the need to open the airway outweighs the risk of further spinal damage in the cardiac arrest patient.
- When spinal injury is suspected or cannot be ruled out, rescuers should maintain manual spinal motion restriction and not use immobilization devices. Manual stabilization can decrease movement of the cervical spine during patient care while allowing for proper ventilation and airway control.23,24 Spinal immobilization devices may make it more difficult to maintain airway patency25,26 and provide adequate ventilation.
This topic last received formal evidence review in 2010.22
Introduction
High-quality CPR is, along with defibrillation for those with shockable rhythms, the most important lifesaving intervention for a patient in cardiac arrest. The evidence for what constitutes optimal CPR continues to evolve as research emerges. A number of key components have been defined for high-quality CPR, including minimizing interruptions in chest compressions, providing compressions of adequate rate and depth, avoiding leaning on the chest between compressions, and avoiding excessive ventilation.1 However, controlled studies are relatively lacking, and observational evidence is at times conflicting. The effect of individual CPR quality metrics or interventions is difficult to evaluate because so many happen concurrently and may interact with each other in their effect. Compression rate and compression depth, for example, have both been associated with better outcomes, yet these variables have been found to be inversely correlated with each other so that improving one may worsen the other.1–3 CPR quality interventions are often applied in “bundles,” making the benefit of any one specific measure difficult to ascertain. As more and more centers and EMS systems are using feedback devices and collecting data on CPR measures such as compression depth and chest compression fraction, these data will enable ongoing updates to these recommendations.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. When providing chest compressions, the rescuer should place the heel of one hand on the center (middle) of the victim’s chest (the lower half of the sternum) and the heel of the other hand on top of the first so that the hands are overlapped. |
| 1 | C-EO | 2. Resuscitation should generally be conducted where the victim is found, as long as high-quality CPR can be administered safely and effectively in that location. |
| 2a | C-LD | 3. It is preferred to perform CPR on a firm surface and with the victim in the supine position, when feasible. |
| 2b | C-LD | 4. When the victim cannot be placed in the supine position, it may be reasonable for rescuers to provide CPR with the victim in the prone position, particularly in hospitalized patients with an advanced airway in place. |
Recommendation-Specific Supportive Text
- A 2020 ILCOR systematic review identified 3 studies involving 57 total patients that investigated the effect of hand positioning on resuscitation process and outcomes.4 Although no difference in resuscitation outcomes was noted, 2 studies found better physiological parameters (peak arterial pressure, mean arterial pressure [MAP], end-tidal carbon dioxide [ETCO2]) when compression was performed over the lower third of the sternum compared with the middle of the sternum.5,6 A third study found no difference.7 Radiographic studies show the left ventricle is typically located inferior to the internipple line, corresponding with the lower half of the sternum.8 However, hand placement inferior to the internipple line may result in compression over the xiphoid.9 Although data from manikin studies conflict, it does not appear to matter whether the dominant or nondominant hand is placed in contact with the sternum.10,11
- The primary considerations when determining if a victim needs to be moved before starting resuscitation are feasibility and safety of providing high-quality CPR in the location and position in which the victim is found. This is a separate question from the decision of if or when to transport a patient to the hospital with resuscitation ongoing.
- The effectiveness of CPR appears to be maximized with the victim in a supine position and the rescuer kneeling beside the victim’s chest (eg, out-of-hospital) or standing beside the bed (eg, in-hospital).12 It is thought that optimal chest compressions are best delivered with the victim on a firm surface.13,14 Manikin studies show generally acceptable thoracic compression with CPR performed on a hospital mattress.
- An older systematic review identified 22 case reports of CPR being performed in the prone position (21 in the operating room, 1 in the intensive care unit [ICU]), with 10/22 patients surviving.15 In a small case series of 6 patients with refractory IHCA, prone positioning with the use of a board with sandbag to compress the sternum improved hemodynamics during CPR but did not result in ROSC.16 The efficacy of CPR in the prone position is not established, but the very limited evidence suggests it may be better than providing no CPR, when a patient cannot be placed in supine position, or until this can be done safely.
Recommendations 1, 2, and 3 are supported by the 2020 CoSTR for BLS.4 Recommendation 4 last received formal evidence review in 2010.17
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. In adult cardiac arrest, total preshock and postshock pauses in chest compressions should be as short as possible. |
| 1 | C-LD | 2. The healthcare provider should minimize the time taken to check for a pulse (no more than 10 s) during a rhythm check, and if the rescuer does not definitely feel a pulse, chest compressions should be resumed. |
| 2a | B-R | 3. When 2 or more rescuers are available, it is reasonable to switch chest compressors approximately every 2 min (or after about 5 cycles of compressions and ventilation at a ratio of 30:2) to prevent decreases in the quality of compressions. |
| 2a | B-R | 4. It is reasonable to immediately resume chest compressions after shock delivery for adults in cardiac arrest in any setting. |
| 2a | C-LD | 5. For adults in cardiac arrest receiving CPR without an advanced airway, it is reasonable to pause compressions to deliver 2 breaths, each given over 1 s. |
| 2b | C-LD | 6. In adult cardiac arrest, it may be reasonable to perform CPR with a chest compression fraction of at least 60%. |
Recommendation-Specific Supportive Text
- Observational evidence suggests improved outcomes with increased chest compression fraction in patients with shockable rhythms.18,19 Specifically, studies have also reported increased ROSC with shorter perishock pauses.20–22
- This recommendation is based on the overall principle of minimizing interruptions to CPR and maintaining a chest compression fraction of at least 60%, which studies have reported to be associated with better outcome.18,19,23
- Chest compression depth begins to decrease after 90 to 120 seconds of CPR, although compression rates do not decrease significantly over that time window.24 A randomized trial using manikins found no difference in the percentage of high-quality compressions when rotating every 1 minute compared with every 2 minutes.25 Rotating the designated chest compressor every 2 minutes is sensible because this approach maintains chest compression quality and takes advantage of when CPR would ordinarily be paused for rhythm analysis.
- Two RCTs enrolling more than 1000 patients did not find any increase in survival when pausing CPR to analyze rhythm after defibrillation.26,27 Observational studies show decreased ROSC when chest compressions are not resumed immediately after shock.28,29
- Because chest compression fraction of at least 60% is associated with better resuscitation outcomes, compression pauses for ventilation should be as short as possible.18,19,23
- A 2015 systematic review reported significant heterogeneity among studies, with some studies, but not all, reporting better rates of survival to hospital discharge associated with higher chest compression fractions.18,19,23 In 2 studies, higher chest compression fraction was associated with lower odds of survival.2,30 Compression rate and depth and cointerventions such as defibrillation, airway management, and medications, are also important and may interact with chest compression fraction. High-performing EMS systems target at least 60%, with 80% or higher being a frequent goal.
Recommendations 1 and 4 are supported by the 2020 CoSTR for BLS.4 Recommendations 2, 3, 5, and 6 last received formal evidence review in 2015.31
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. During manual CPR, rescuers should perform chest compressions to a depth of at least 2 inches, or 5 cm, for an average adult while avoiding excessive chest compression depths (greater than 2.4 inches, or 6 cm). |
| 2a | B-NR | 2. In adult victims of cardiac arrest, it is reasonable for rescuers to perform chest compressions at a rate of 100 to 120/min. |
| 2a | C-LD | 3. It can be beneficial for rescuers to avoid leaning on the chest between compressions to allow complete chest wall recoil for adults in cardiac arrest. |
| 2b | C-EO | 4. It may be reasonable to perform chest compressions so that chest compression and recoil/relaxation times are approximately equal. |
Recommendation-Specific Supportive Text
- A 2020 ILCOR scoping review32 identified 12 studies, including over 12 500 patients, looking at chest compression components. Several studies found better outcomes, including survival to hospital discharge and defibrillation success, when compression depth was at least 5 cm compared with less than 4 cm.3,20,33,34
- The same review32 identified 13 studies, involving 15 000 patients, that looked at compression rate. Results were somewhat inconsistent across studies, with only 3 observational studies in adults showing an association between higher compression rate and outcomes.1,35,36 The only RCT identified included 292 patients and compared a rate of 100 to a rate of 120, finding no difference in outcomes.37 There is no evidence to suggest altering the suggested compression rate of 100 to 120/min in adults. Three studies have reported that depth decreases as rate increases, highlighting the pitfalls of evaluating a single CPR quality metric in isolation.1–3
- The ILCOR review32 identified 2 observational studies that provided inconsistent results on the association between chest compression release velocity and survival, with 1 study finding no association and the other finding that faster release velocity was associated with increased survival.38,39 Not allowing complete chest wall recoil has been associated with increased intrathoracic pressure and decreased coronary perfusion.40,41
- CPR duty cycle refers to the proportion of time spent in compression relative to the total time of the compression plus decompression cycle. The 2010 Guidelines recommended a 50% duty cycle, in which the time spent in compression and decompression was equal, mainly on the basis of its perceived ease of being achieved in practice. Notably, in a clinical study in adults with out-of- hospital VF arrest (of whom 43% survived to hospital discharge), the mean duty cycle observed during resuscitation was 39%.42 A study in children also found the mean duty cycle was 40%, suggesting that shorter duty cycles may be the norm in clinical practice.43 Although many animal studies have observed higher blood flows and better outcomes when the duty cycle was less than 50%, the optimal duty cycle is not known. Currently, there is insufficient evidence to warrant a change from the existing recommendation, which remains a knowledge gap that requires further investigation.
Recommendations 1, 2, and 3 are supported by the 2020 CoSTR for BLS.4 Recommendation 4 last received formal evidence review in 2010.44
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-R | 1. It may be reasonable to use audiovisual feedback devices during CPR for real-time optimization of CPR performance. |
| 2b | C-LD | 2. It may be reasonable to use physiological parameters such as arterial blood pressure or end-tidal CO2 when feasible to monitor and optimize CPR quality. |
Recommendation-Specific Supportive Text
- A 2020 ILCOR systematic review found that most studies did not find a significant association between real-time feedback and improved patient outcomes.4 However, no studies identified significant harm, and some demonstrated clinically important improvement in survival. One recent RCT reported a 25.6% increase in survival to hospital discharge from IHCA with audio feedback on compression depth and recoil (54% versus 28.4%; P<0.001).45
- An analysis of data from the AHA’s Get With The Guidelines-Resuscitation registry showed higher likelihood of ROSC (odds ratio, 1.22; 95% CI, 1.04–1.34; P=0.017) when CPR quality was monitored using either ETCO2 or diastolic blood pressure.46 An observational study in adult patients (IHCA and OHCA) reported that for every 10 mm compression depth increase, ETCO2 increased 1.4 mm Hg.47 A 2018 systematic review of ETCO2 as a prognostic indicator for ROSC48 found variability in cutoff values, but less than 10 mm Hg was generally associated with poor outcome and greater than 20 mm Hg had a stronger association with ROSC than a value of greater than 10 mm Hg. The combination of the association of higher ETCO2 with ROSC and the finding that increased chest compression depth can increase ETCO2 suggests that targeting compressions to a value of at least 10 mm Hg, and ideally 20 mm Hg or greater, may be useful. The validity and reliability of ETCO2 in nonintubated patients is not well established. When available, invasive arterial blood pressure monitoring may also help assess and guide CPR efforts. The use of diastolic blood pressure monitoring during cardiac arrest was associated with higher ROSC,46 but there are inadequate human data to suggest any specific pressure.
These recommendations are supported by the 2020 CoSTRs for BLS and ALS.4,49
Introduction
The provision of rescue breaths for apneic patients with a pulse is essential. The relative contribution of assisted ventilation for patients in cardiac arrest is more controversial.
There is concern that delivery of chest compressions without assisted ventilation for prolonged periods could be less effective than conventional CPR (compressions plus breaths) because the arterial oxygen content will decrease as CPR duration increases. This concern is especially pertinent in the setting of asphyxial cardiac arrest. Much of the published research involves patients whose arrests were presumed to be of cardiac origin and in settings with short EMS response times. It is likely that a time threshold exists beyond which the absence of ventilation may be harmful, and the generalizability of the findings to all settings must be considered with caution.1
Once an advanced airway has been placed, delivering continuous chest compressions increases the compression fraction but makes it more difficult to deliver adequate ventilation. Simultaneous compressions and ventilation should be avoided,2 but delivery of chest compressions without pausing for ventilation seems a reasonable option.3 The use of SGAs adds to this complexity because efficiency of ventilation during cardiac arrest may be worse than when using an endotracheal tube, though this has not been borne out in recently published RCTs.4,5
| COR | LOE | Recommendations |
|---|---|---|
| 2a | C-LD | 1. For adults in cardiac arrest receiving ventilation, tidal volumes of approximately 500 to 600 mL, or enough to produce visible chest rise, are reasonable. |
| 2a | C-EO | 2. In patients without an advanced airway, it is reasonable to deliver breaths either by mouth or by using bag-mask ventilation. |
| 2b | C-EO | 3. When providing rescue breaths, it may be reasonable to give 1 breath over 1 s, take a “regular” (not deep) breath, and give a second rescue breath over 1 s. |
| 3: Harm | C-LD | 4. Rescuers should avoid excessive ventilation (too many breaths or too large a volume) during CPR. |
Recommendation-Specific Supportive Text
- Studies have reported that enough tidal volume to cause visible chest rise, or approximately 500 to 600 mL, provides adequate ventilation while minimizing the risk of overdistension or gastric insufflation.6–9
- Both mouth-to-mouth rescue breathing and bagmask ventilation provide oxygen and ventilation to the victim.10 To provide mouth-to-mouth rescue breaths, open the victim’s airway, pinch the victim’s nose, create an airtight mouth-to-mouth seal, and provide a breath.
- Taking a regular rather than a deep breath prevents the rescuer from getting dizzy or lightheaded and prevents overinflation of the victim’s lungs. The most common cause of ventilation difficulty is an improperly opened airway,11 so if the victim’s chest does not rise with the first rescue breath, reposition the head by performing the head tilt–chin lift again and then give the second rescue breath. The recommendation for 1 second is to keep the pauses in CPR as brief as possible.
- Excessive ventilation is unnecessary and can cause gastric inflation, regurgitation, and aspiration.12,14 Excessive ventilation can also be harmful by increasing intrathoracic pressure, decreasing venous return to the heart, and diminishing cardiac output and survival.14
This topic last received formal evidence review in 2010.15
| COR | LOE | Recommendations |
|---|---|---|
| 2a | C-LD | 1. It is reasonable for a rescuer to use mouth-to-nose ventilation if ventilation through the victim’s mouth is impossible or impractical. |
| 2b | C-EO | 2. For a victim with a tracheal stoma who requires rescue breathing, either mouth-to-stoma or face mask (pediatric preferred)– to–stoma ventilation may be reasonable. |
Recommendation-Specific Supportive Text
- Mouth-to-nose ventilation may be necessary if ventilation through the victim’s mouth is impossible because of trauma, positioning, or difficulty obtaining a seal. A case series suggests that mouth-to-nose ventilation in adults is feasible, safe, and effective.16
- Effective ventilation of the patient with a tracheal stoma may require ventilation through the stoma, either by using mouth-to-stoma rescue breaths or by use of a bag-mask technique that creates a tight seal over the stoma with a round, pediatric face mask. There is no published evidence on the safety, effectiveness, or feasibility of mouth-to-stoma ventilation. One study of patients with laryngectomies showed that a pediatric face mask created a better peristomal seal than a standard ventilation mask.17
This topic last received formal evidence review in 2010.15
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. If an adult victim with spontaneous circulation (ie, strong and easily palpable pulses) requires support of ventilation, it may be reasonable for the healthcare provider to give rescue breaths at a rate of about 1 breath every 6 s, or about 10 breaths per minute. |
Recommendation-Specific Supportive Text
- Since the last review in 2010 of rescue breathing in adult patients, there has been no evidence to support a change in previous recommendations. A study in critically ill patients who required ventilatory support found that bag-mask ventilation at a rate of 10 breaths per minute decreased hypoxic events before intubation.18
This topic last received formal evidence review in 2010.15
| COR | LOE | Recommendations |
|---|---|---|
| 2a | B-R | 1. Before placement of an advanced airway (supraglottic airway or tracheal tube), it is reasonable for healthcare providers to perform CPR with cycles of 30 compressions and 2 breaths. |
| 2b | B-R | 2. It may be reasonable for EMS providers to use a rate of 10 breaths per minute (1 breath every 6 s) to provide asynchronous ventilation during continuous chest compressions before placement of an advanced airway. |
| 2b | C-LD | 3. If an advanced airway is in place, it may be reasonable for the provider to deliver 1 breath every 6 s (10 breaths/min) while continuous chest compressions are being performed. |
| 2b | C-LD | 4. It may be reasonable to initially use minimally interrupted chest compressions (ie, delayed ventilation) for witnessed shockable OHCA as part of a bundle of care. |
Recommendation-Specific Supportive Text
- A 2017 ILCOR systematic review found that a ratio of 30 compressions to 2 breaths was associated with better survival than alternate ratios, a recommendation that was reaffirmed by the AHA in 2018.19,20 Most of these studies examined “bundles” of cardiac arrest care, making it impossible to know if the improvement was due to the compression-to-ventilation ratio itself. This ratio is supported by a large OHCA RCT in which the use of 30:2 (with a pause in compressions of less than 5 seconds) was at least as good as continuous chest compressions.21
- In a large trial, survival and survival with favorable neurological outcome were similar in a group of patients with OHCA treated with ventilations at a rate of 10/min without pausing compressions, compared with a 30:2 ratio before intubation.21
- A 2017 systematic review identified 1 observational human study and 10 animal studies comparing different ventilation rates after advanced airway placement.22 No clear benefit from a rate of 10 was identified, but no other rate was found to be superior. A 2017 ILCOR systematic review did not identify any new evidence to alter this recommendation, which was reiterated in the “2017 AHA Focused Update on Adult BLS and CPR Quality: An Update to the AHA Guidelines for CPR and Emergency Cardiovascular Care.”19,20
- A 2017 ILCOR systematic review concluded that although the evidence from observational studies supporting the use of bundles of care including minimally interrupted chest compressions was of very low certainty (primarily unadjusted results), systems already using such an approach may continue to do so.19
These recommendations are supported by the 2017 focused update on adult BLS and CPR quality guidelines.20
Introduction
Along with CPR, early defibrillation is critical to survival when sudden cardiac arrest is caused by VF or pulseless VT (pVT).1,2 Defibrillation is most successful when administered as soon as possible after onset of VF/VT and a reasonable immediate treatment when the interval from onset to shock is very brief. Conversely, when VF/ VT is more protracted, depletion of the heart’s energy reserves can compromise the efficacy of defibrillation unless replenished by a prescribed period of CPR before the rhythm analysis. Minimizing disruptions in CPR surrounding shock administration is also a high priority.
Currently marketed defibrillators use proprietary shock waveforms that differ in their electric characteristics. These deliver different peak currents even at the same programmed energy setting, making comparisons of shock efficacy between devices challenging. Energy setting specifications for cardioversion also differ between defibrillators. Refer to the device manufacturer’s recommended energy for a particular waveform.
Technologies are now in development to diagnose the underlying cardiac rhythm during ongoing CPR and to derive prognostic information from the ventricular waveform that can help guide patient management. These still require further testing and validation before routine use.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. Defibrillators (using biphasic or monophasic waveforms) are recommended to treat tachyarrhythmias requiring a shock. |
| 2a | B-R | 2. Based on their greater success in arrhythmia termination, defibrillators using biphasic waveforms are preferred over monophasic defibrillators for treatment of tachyarrhythmias. |
| 2a | B-NR | 3. A single shock strategy is reasonable in preference to stacked shocks for defibrillation in the setting of unmonitored cardiac arrest. |
| 2a | C-LD | 4. It is reasonable that selection of fixed versus escalating energy levels for subsequent shocks for presumed shock-refractory arrhythmias be based on the specific manufacturer’s instructions for that waveform. If this is not known, defibrillation at the maximal dose may be considered. |
| 2b | B-R | 5. If using a defibrillator capable of escalating energies, higher energy for second and subsequent shocks may be considered for presumed shock-refractory arrhythmias. |
| 2b | C-LD | 6. In the absence of conclusive evidence that one biphasic waveform is superior to another in termination of VF, it is reasonable to use the manufacturer’s recommended energy dose for the first shock. If this is not known, defibrillation at the maximal dose may be considered. |
Recommendation-Specific Supportive Text
- Emergent electric cardioversion and defibrillation are highly effective at terminating VF/VT and other tachyarrhythmias. No shock waveform has distinguished itself as achieving a consistently higher rate of ROSC or survival. Biphasic and monophasic shock waveforms are likely equivalent in their clinical outcome efficacy.3
- No shock waveform has proved to be superior in improving the rate of ROSC or survival. However, biphasic waveform defibrillators (which deliver pulses of opposite polarity) expose patients to a much lower peak electric current with equivalent or greater efficacy for terminating atrial4 and ventricular tachyarrhythmias than monophasic (single polarity) defibrillators do.5–10,13 These potential differences in safety and efficacy favor preferential use of a biphasic defibrillator, when available Biphasic defibrillators have largely replaced monophasic shock defibrillators, which are no longer manufactured.
- The rationale for a single shock strategy, in which CPR is immediately resumed after the first shock rather than after serial “stacked” shocks (if required) is based on a number of considerations. These include the high success rate of the first shock with biphasic waveforms (lessening the need for successive shocks), the declining success of immediate second and third serial shocks when the first shock has failed,14 and the protracted interruption in CPR required for a series of stacked shocks. A single shock strategy results in shorter interruptions in CPR and a significantly improved survival to hospital admission and discharge (although not 1-year survival) compared with serial “stacked” shocks.15–17 It is unknown whether stacked shocks or single shocks are more effective in settings of a monitored witnessed arrest (for example, see the section on Cardiac Arrest After Cardiac Surgery).
- Regardless of waveform, successful defibrillation requires that a shock be of sufficient energy to terminate VF/VT. In cases where the initial shock fails to terminate VF/VT, subsequent shocks may be effective when repeated at the same or an escalating energy setting.18,19 An optimal energy setting for first or subsequent biphasic defibrillation, whether fixed or escalating, has not been identified, and its selection can be based on the defibrillator’s manufacturer specification.
- There is no conclusive evidence of superiority of one biphasic shock waveform over another for defibrillation.20 Given the variability in electric characteristics between proprietary biphasic waveforms, it is reasonable to use the energy settings specified by the manufacturer for that specific device. If a manufacturer’s specified energy setting for defibrillation is not known at the time of intended use, the maximum dose setting for that device may be considered.
- Commercially available defibrillators either provide fixed energy settings or allow for escalating energy settings; both approaches are highly effective in terminating VF/VT.18 An optimal energy setting for first or subsequent biphasic defibrillation, whether fixed or escalating, has not been identified and is best deferred to the defibrillator’s manufacturer. A randomized trial comparing fixed 150 J biphasic defibrillation with escalating higher shock energies (200–300–360 J) observed similar rates of successful defibrillation and conversion to an organized rhythm after the first shock. However, among patients who required multiple shocks, escalating shock energy resulted in a significantly higher rate of conversion to an organized rhythm, although overall survival did not differ between the 2 treatment groups.19 When VF/VT is refractory to the first shock, an equivalent or higher energy setting than the first shock may be considered. As yet, there is no conclusive evidence of superiority of one biphasic shock waveform over another for defibrillation.20 It is reasonable to use the energy settings specified by the manufacturer for that specific device. If a manufacturer’s specified energy setting for defibrillation is not known at the time of intended use, the maximum dose setting for that device may be considered.
Recommendations 1, 2, and 6 last received formal evidence review in 2015.21 Recommendations 3, 4, and 5 are supported by the 2020 CoSTR for BLS.22
| COR | LOE | Recommendation |
|---|---|---|
| 2a | C-LD | 1. It is reasonable to place defibrillation paddles or pads on the exposed chest in an anterolateral or anteroposterior position, and to use a paddle or pad electrode diameter more than 8 cm in adults. |
Recommendation-Specific Supportive Text
- Anterolateral, anteroposterior, anterior-left infrascapular, and anterior-right infrascapular electrode placements are comparably effective for treating supraventricular and ventricular arrhythmias. 24-28 A larger pad/paddle size (within the limits of 8-12 cm in diameter) lowers transthoracic impedance.29,30 Self-adhesive pads have largely replaced defibrillation paddles in clinical practice. Before pad placement, remove all clothing and jewelry from the chest.
This recommendation is supported by a 2020 ILCOR scoping review, which found no new information to update the 2010 recommendations.22,31
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. It may be reasonable to use a defibrillator in manual mode as compared with automatic mode depending on the skill set of the operator. |
Recommendation-Specific Supportive Text
- AEDs are highly accurate in their detection of shockable arrhythmias but require a pause in CPR for automated rhythm analysis.32,33 Manual defibrillation can result in a shorter hands-off period for rhythm confirmation in operators with a sufficient skill for rapid and reliable rhythm interpretation.34,35
This recommendation is supported by a 2020 ILCOR scoping review,22 which found no new information to update the 2010 recommendations.31
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. CPR is recommended until a defibrillator or AED is applied. |
| 2a | B-R | 2. In unmonitored cardiac arrest, it is reasonable to provide a brief prescribed period of CPR while a defibrillator is being obtained and readied for use before initial rhythm analysis and possible defibrillation. |
| 2a | C-LD | 3. Immediate defibrillation is reasonable for provider-witnessed or monitored VF/pVT of short duration when a defibrillator is already applied or immediately available. |
Recommendation-Specific Supportive Text
- CPR is the single-most important intervention for a patient in cardiac arrest and should be provided until a defibrillator is applied to minimize interruptions in compressions.
- When VF/VT has been present for more than a few minutes, myocardial reserves of oxygen and other energy substrates are rapidly depleted. If replenished by a period of CPR before shock, defibrillation success improves significantly.1,2,36,37 Because no differences in outcome were seen in studies comparing short (typically approximately about 30 seconds) with prolonged (up to 3 minutes) periods of CPR preceding the initial rhythm analysis, a brief period of CPR while the defibrillator is readied for use may be sufficient in unmonitored cardiac arrest.38–40 Even in monitored arrests, it can take time to attach pads, power on a defibrillator, and charge the capacitor before shock delivery, during which there is good reason to administer CPR.
- Early defibrillation improves outcome from cardiac arrest.41–43 When VF is of short duration, myocardial reserves of oxygen and other energy substrates are likely to remain intact. During this early electric phase, the rhythm is most responsive to defibrillation.44,45 Thus, if the onset of VF is monitored or witnessed with a defibrillator that is already applied, or to which there is immediate access, it is reasonable to administer a shock as soon as possible. Interim CPR should be provided if there is any delay in obtaining or readying the defibrillator for use.
Recommendations 1 and 2 are supported by the 2020 CoSTR for BLS.22 Recommendation 3 last received formal evidence review in 2010.46
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-EO | 1. It may be reasonable to charge a manual defibrillator during chest compressions either before or after a scheduled rhythm analysis. |
Recommendation-Specific Supportive Text
- There are differing approaches to charging a manual defibrillator during resuscitation. It is not uncommon for chest compressions to be paused for rhythm detection and continue to be withheld while the defibrillator is charged and prepared for shock delivery. This approach results in a protracted hands-off period before shock. Precharging the defibrillator during ongoing chest compressions shortens the hands-off chest time surrounding defibrillation, without evidence of harm.47 Although no study has directly evaluated the effect of precharging itself on cardiac arrest outcome, shorter perishock pauses (which could result from such a strategy) are associated with improved survival from VF arrest.48 Two approaches are reasonable: either charging the defibrillator before a rhythm check or resuming compressions briefly after a rhythm check while the defibrillator charges. Either approach may reduce no-flow time.49,50
This recommendation is supported by the 2020 CoSTR for ALS.51
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. It may be reasonable to immediately resume chest compressions after shock administration rather than pause CPR to perform a postshock rhythm check in cardiac arrest patients. |
Recommendation-Specific Supportive Text
-
Immediate resumption of chest compressions after shock results in a shorter perishock pause and improves the overall hands-on time (chest compression fraction) during resuscitation, which is associated with improved survival from VF arrest.16,48 Even when successful, defibrillation is often followed by a variable (and sometimes protracted) period of asystole or pulseless electrical activity, during which providing CPR while awaiting a return of rhythm and pulse is advisable. Whether resumption of CPR immediately after shock might reinduce VF/VT is controversial.52-54 This potential concern has not been borne out by any evidence of worsened survival from such a strategy. Should there be physiological evidence of return of circulation such as an arterial waveform or abrupt rise in ETCO2 after shock, a pause of chest compressions briefly for confirmatory rhythm analysis may be warranted.
This recommendation is supported by the 2020 CoSTR for BLS.22
| COR | LOE | Recommendations |
|---|---|---|
| 2b | C-LD | 1. The value of artifact-filtering algorithms for analysis of electrocardiogram (ECG) rhythms during chest compressions has not been established. |
| 2b | C-LD | 2. The value of VF waveform analysis to guide the acute management of adults with cardiac arrest has not been established. |
Recommendation-Specific Supportive Text
- CPR obscures interpretation of the underlying rhythm because of the artifact created by chest compressions on the ECG. This makes it difficult to plan the next step of care and can potentially delay or even misdirect drug therapies if given empirically (blindly) based on the patient’s presumed, but not actual, underlying rhythm. Time taken for rhythm analysis also disrupts CPR. Artifact-filtering and other innovative techniques to disclose the underlying rhythm beneath ongoing CPR can surmount these challenges and minimize interruptions in chest compressions while offering a diagnostic advantage to better direct therapies.55–60 Despite the theoretical advantages, no study has evaluated these technologies in a real-time clinical setting or validated their clinical effectiveness compared to current resuscitation strategies. At present, filtering algorithms are strictly used for visual (manual) rhythm interpretation and not for automated VF/VT rhythm detection in AEDs during ongoing CPR. This added potential application remains untested. Recognizing the need for further clinical research, a 2020 ILCOR systematic review recommended against adopting artifact-filtering algorithms for rhythm analysis during CPR at the present time.51 The writing group also endorses the need for further investigation and clinical validation before these technologies are adopted into clinical practice.
- The electric characteristics of the VF waveform are known to change over time.61 VF waveform analysis may be of value in predicting the success of defibrillation or other therapies during the course of resuscitation.62–64 The prospect of basing therapies on a prognostic analysis of the VF waveform in real-time is an exciting and developing avenue of new research. However, the validity, reliability, and clinical effectiveness of an approach that prompts or withholds shock or other therapies on the basis of predictive analyses is currently uncertain. The only prospective clinical trial comparing a standard shock-first protocol with a waveform analysis-guided shock algorithm observed no differences in outcome.65 The consensus of the writing group is that there is currently insufficient evidence to support the routine use of waveform analysis to guide resuscitation care, but it is an area in which further research with clinical validation is needed and encouraged.
Recommendation 1 is supported by the 2020 CoSTR for ALS.51 Recommendation 2 is supported by a 2020 ILCOR evidence update,51 which found no new information to update the 2010 recommendations.66
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. The usefulness of double sequential defibrillation for refractory shockable rhythm has not been established. |
Recommendation-Specific Supportive Text
- There is limited evidence examining double sequential defibrillation in clinical practice. A number of case reports have shown good outcomes in patients who received double sequential defibrillation. However, these case reports are subject to publication bias and should not be used to support its effectiveness.67 A handful of observational studies demonstrated no difference in outcomes (ROSC, survival, neurological outcome) with the use of double sequential defibrillation compared with standard defibrillation.68–71 These studies should also be interpreted with caution, because the use of double sequential defibrillation was not protocolized and was often used late in the resuscitation after standard resuscitation was unsuccessful. Published reports also do not distinguish the application of double sequential defibrillation for truly shock-refractory (incessant) VF versus VF that recurs during the period of CPR after a successful shock, which is the more common clinical scenario.3,7 A 2020 ILCOR systematic review found no evidence to support double sequential defibrillation and recommended against its routine use compared with standard defibrillation.51 A recent pilot RCT (not included in the systematic review) of 152 patients who remained in VF after at least 3 shocks found higher rates of VF termination and ROSC with double sequential defibrillation or alternative defibrillator pad placement compared with standard defibrillation but was not powered for these outcomes and did not report patient survival.72 A number of unanswered questions remain about double sequential defibrillation, including intershock timing, pad positioning, technique, and the possibility of harm with increased energy and defibrillator damage.73,74 It is premature for double sequential defibrillation to be incorporated into routine clinical practice given the lack of evidence. Its usefulness should be explored in the context of clinical trials. An ongoing RCT (NCT04080986) may provide answers to some of these questions.
This recommendation is supported by the 2020 CoSTR for ALS.51
Introduction
In addition to defibrillation, several alternative electric and pseudoelectrical therapies have been explored as possible treatment options during cardiac arrest. Transcutaneous pacing has been studied during cardiac arrest with bradyasystolic cardiac rhythm. The theory is that the heart will respond to electric stimuli by producing myocardial contraction and generating forward movement of blood, but clinical trials have not shown pacing to improve patient outcomes.
Other pseudoelectrical therapies, such as cough CPR, fist or percussion pacing, and precordial thump have all been described as temporizing measures in select patients who are either periarrest or in the initial seconds of witnessed cardiac arrest (before losing consciousness in the case of cough CPR) when definitive therapy is not readily available. Precordial thump is a single, sharp, high-velocity impact (or “punch”) to the middle sternum by the ulnar aspect of a tightly clenched fist. The force from a precordial thump is intended to transmit electric energy to the heart, similar to a low-energy shock, in hope of terminating the underlying tachyarrhythmia.
Fist (or percussion) pacing is the delivery of a serial, rhythmic, relatively low-velocity impact to the sternum by a closed fist.1 Fist pacing is administered in an attempt to stimulate an electric impulse sufficient to cause myocardial depolarization. Cough CPR is described as repeated deep breaths followed immediately by a cough every few seconds in an attempt to increase aortic and intracardiac pressures, providing transient hemodynamic support before a loss of consciousness.
| COR | LOE | Recommendation |
|---|---|---|
| 3: No Benefit | B-R | 1. Electric pacing is not recommended for routine use in established cardiac arrest. |
Recommendation-Specific Supportive Text
- Existing evidence, including observational and quasi-RCT data, suggests that pacing by a transcutaneous, transvenous, or transmyocardial approach in cardiac arrest does not improve the likelihood of ROSC or survival, regardless of the timing of pacing administration in established asystole, location of arrest (in-hospital or out-of-hospital), or primary cardiac rhythm (asystole, pulseless electrical activity).2–6 Protracted interruptions in chest compressions while the success of pacing is assessed can also be detrimental to survival. It is not known whether the timing of pacing initiation may influence pacing success such that pacing may be useful in the initial seconds of select cases of witnessed, monitored cardiac arrest (see the section on Cardiac Arrest After Cardiac Surgery). If pacing is attempted during cardiac arrest related to the special circumstances described above, providers are cautioned against its performance at the expense of high-quality CPR, particularly when assessing electric and mechanical capture.
This topic last underwent formal evidence review in 2010.7
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. The precordial thump may be considered at the onset of a rescuer-witnessed, monitored, unstable ventricular tachyarrhythmia when a defibrillator is not immediately ready for use and is performed without delaying CPR or shock delivery. |
| 3: No Benefit | C-LD | 2. The precordial thump should not be used routinely for established cardiac arrest. |
Recommendation-Specific Supportive Text
- and 2. The intent of precordial thump is to transmit the mechanical force of the “thump” to the heart as electric energy analogous to a pacing stimulus or very low-energy shock (depending on its force) and is referred to as electromechanical transduction.1 There is no evidence that the use of precordial thump during routine cardiac arrest care in the out-of-hospital or in-hospital settings improves rates of ROSC or survival to hospital discharge.8–12 It may be beneficial only at the very early onset of VT when the arrhythmia is most vulnerable to lower-energy termination such as in responder-witnessed, monitored events, or in a controlled laboratory environment, but even then it is rarely effective.13 Although there are case reports of success without evidence of harm from a precordial thump,9,14,15 if fortuitously administered on the electrically vulnerable portion of an organized rhythm (T wave), the thump (like an unsynchronized shock) risks acceleration or conversion of the rhythm to VF,16–19 analogous to commotio cordis.20 Thus, although the thump may be useful as a single brief intervention under specific circumstances (ie, when a cardiac arrest is witnessed by the responder and monitor-confirmed to be due to VF/VT and a defibrillator is not readily available for use), it should not delay CPR or deployment of a defibrillator.
These recommendations are supported by the 2020 CoSTR for BLS.21
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. Fist (percussion) pacing may be considered as a temporizing measure in exceptional circumstances such as witnessed, monitored in-hospital arrest (eg, cardiac catheterization laboratory) for bradyasystole before a loss of consciousness and if performed without delaying definitive therapy. |
Recommendation-Specific Supportive Text
- Fist, or percussion, pacing is administered with the goal of stimulating an electric impulse sufficient to cause depolarization and contraction of the myocardium, resulting in a pulse. There are a number of case reports and case series that examined the use of fist pacing during asystolic or “life-threatening bradycardic” events1,22–25 showing favorable outcomes of survival22 and ROSC.23 None of these studies, however, were controlled or comparative, and it is not known if the use of fist pacing itself improves rates of ROSC or survival compared with standard therapy. There is no role for fist pacing in patients in cardiac arrest.
This recommendation is supported by the 2020 CoSTR for BLS.21
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. “Cough” CPR may be considered as a temporizing measure for the witnessed, monitored onset of a hemodynamically significant tachyarrhythmia or bradyarrhythmia before a loss of consciousness without delaying definitive therapy. |
Recommendation-Specific Supportive Text
- It is important to underscore that while cough CPR by definition cannot be used for an unconscious patient, it can be harmful in any setting if diverting time, effort, and attention from performing high-quality CPR. Cough CPR is described as a repetitive deep inspiration followed by a cough every few seconds before the loss of consciousness. It is feasible only at the onset of a hemodynamically significant arrhythmia in a cooperative, conscious patient who has ideally been previously instructed on its performance, and as a bridge to definitive care. There are no studies comparing cough CPR to standard resuscitation care. Limited evidence from case reports and case series demonstrates transient increases in aortic and intracardiac pressure with the use of cough CPR at the onset of tachyarrhythmias or bradyarrhythmias in conscious patients.10,26–28 These studies suffer from considerable selection bias and lack of comparison groups, and do not control for the confounding effect of other treatments, making them hard to interpret.
This recommendation is supported by the 2020 CoSTR for BLS.21
| COR | LOE | Recommendations |
|---|---|---|
| 2a | B-NR | 1. It is reasonable for providers to first attempt establishing intravenous access for drug administration in cardiac arrest. |
| 2b | B-NR | 2. Intraosseous access may be considered if attempts at intravenous access are unsuccessful or not feasible. |
| 2b | C-LD | 3. In appropriately trained providers, central venous access may be considered if attempts to establish intravenous and intraosseous access are unsuccessful or not feasible. |
| 2b | C-LD | 4. Endotracheal drug administration may be considered when other access routes are not available. |
Synopsis
The traditional approach for giving emergency pharmacotherapy is by the peripheral IV route. However, obtaining IV access under emergent conditions can prove to be challenging based on patient characteristics and operator experience leading to delay in pharmacological treatments.
Alternatives to IV access for acute drug administration include IO, central venous, intracardiac, and endotracheal routes. Intracardiac drug administration was discouraged in the 2000 AHA Guidelines for CPR and Emergency Cardiovascular Care given its highly specialized skill set, potential morbidity, and other available options for access.1,2 Endotracheal drug administration results in low blood concentrations and unpredictable pharmacological effect and has also largely fallen into disuse given other access options. Central venous access is primarily used in the hospital setting because it requires appropriate training to acquire and maintain the needed skill set.
IO access has grown in popularity given the relative ease and speed with which it can be achieved, a higher successful placement rate compared with IV cannulation, and the relatively low procedural risk. However, the efficacy of IV versus IO drug administration in cardiac arrest remains to be elucidated.
Recommendation-Specific Supportive Text
- The peripheral IV route has been the traditional approach to vascular access for emergency drug and fluid administration during resuscitation. The pharmacokinetic properties, acute effects, and clinical efficacy of emergency drugs have primarily been described when given intravenously.3–6 The IV route has precedence, is usually accessible, and affords a potentially more predictable drug response, making it a reasonable initial approach for vascular access.
- The paucity of information on the efficacy of IO drug administration during CPR was acknowledged in 2010, but since then the IO route has grown in popularity. IO access is increasingly implemented as a first-line approach for emergent vascular access. A 2020 ILCOR systematic review7 comparing IV versus IO (principally pretibial placement) drug administration during cardiac arrest found the IV route was associated with better clinical outcomes compared with IO in 5 retrospective studies.8–12 There were significant concerns for bias, particularly due to the fact that need for IO placement may indicate patient or arrest characteristics that are also risk factors for poor outcome. Subgroup analyses of IV versus IO route from 2 RCTs were also included in this systematic review. In these, no statistically significant effect modification by route of administration was identified. Point estimates favored IV access except for the outcome of ROSC in the PARAMEDIC2 trial, where the effect of epinephrine was similar regardless of route.13,14 Site specificity may also be an issue with IO administration, because IO access was nearly always pretibial in these studies. On the basis of these results, the writing group concluded that establishing a peripheral IV remains a reasonable initial approach, but IO access may be considered when an IV is not successful or feasible. Further research is needed to assess the efficacy of drugs delivered intravenously as compared with intraosseously (tibial and humeral).
- Drug administration by central venous access (by internal jugular or subclavian vein) achieves higher peak concentrations and more rapid circulation times than drugs administered by peripheral IV do,15–17 but there are currently no data comparing clinical outcomes between these access routes. Central access is associated with higher morbidity, takes time to perform, and may also require interruption of CPR. Current use of this approach is largely in the hospital and may be considered by skilled providers when IV and IO access are not successful or feasible.
- Endotracheal drug administration is regarded as the least-preferred route of drug administration because it is associated with unpredictable (but generally low) drug concentrations18–20 and lower rates of ROSC and survival.21
Recommendations 1 and 2 are supported by the 2020 CoSTR for ALS.22 Recommendations 3 and 4 last received formal evidence review in 2010.20
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-R | 1. We recommend that epinephrine be administered for patients in cardiac arrest. |
| 2a | B-R | 2. Based on the protocols used in clinical trials, it is reasonable to administer epinephrine 1 mg every 3 to 5 min for cardiac arrest. |
| 2a | C-LD | 3. With respect to timing, for cardiac arrest with a nonshockable rhythm, it is reasonable to administer epinephrine as soon as feasible. |
| 2b | C-LD | 4. With respect to timing, for cardiac arrest with a shockable rhythm, it may be reasonable to administer epinephrine after initial defibrillation attempts have failed. |
| 2b | C-LD | 5. Vasopressin alone or vasopressin in combination with epinephrine may be considered in cardiac arrest but offers no advantage as a substitute for epinephrine in cardiac arrest. |
| 3: No Benefit | B-R | 6. High-dose epinephrine is not recommended for routine use in cardiac arrest. |
Synopsis
Epinephrine has been hypothesized to have beneficial effects during cardiac arrest primarily because of its α-adrenergic effects, leading to increased coronary and cerebral perfusion pressure during CPR. Conversely, the β-adrenergic effects may increase myocardial oxygen demand, reduce subendocardial perfusion, and may be proarrhythmic. Two randomized, placebo-controlled trials, enrolling over 8500 patients, evaluated the efficacy of epinephrine for OHCA.1,2 A systematic review and meta-analysis of these and other studies3 concluded that epinephrine significantly increased ROSC and survival to hospital discharge. Epinephrine did not lead to increased survival with favorable or unfavorable neurological outcome at 3 months, although both of these outcomes occurred slightly more frequently in the epinephrine group.2 Observational data suggest better outcomes when epinephrine is given sooner, and the low survival with favorable neurological outcome in the available trials may be due in part to the median time of 21 minutes from arrest to receipt of epinephrine. This time delay is a consistent issue in OHCA trials. Time to drug in IHCA is generally much shorter, and the effect of epinephrine on outcomes in the IHCA population may therefore be different. No trials to date have found any benefit of either higher-dose epinephrine or other vasopressors over standard-dose epinephrine during CPR.
Recommendation-Specific Supportive Text
- The suggestion to administer epinephrine was strengthened to a recommendation based on a systematic review and meta-analysis,3 which included results of 2 randomized trials of epinephrine for OHCA, 1 of which included over 8000 patients,1,2 showing that epinephrine increased ROSC and survival. At 3 months, the time point felt to be most meaningful for neurological recovery, there was a nonsignificant increase in survivors with both favorable and unfavorable neurological outcome in the epinephrine group.2 Any drug that increases the rate of ROSC and survival, but is given after several minutes of downtime, will likely increase both favorable and unfavorable neurological outcome. Determining the likelihood of favorable or unfavorable neurological outcome at the time of arrest is currently not feasible. Therefore, continuing to use a drug that has been shown to increase survival, while focusing our broader efforts on shortening time to drug for all patients so that more survivors will have a favorable neurological outcome, seems the most beneficial approach.
- The existing trials have used a protocol of 1 mg every 3 to 5 minutes. Operationally, administering epinephrine every second cycle of CPR, after the initial dose, may also be reasonable.
- Of 16 observational studies on timing in the recent systematic review, all found an association between earlier epinephrine and ROSC for patients with nonshockable rhythms, although improvements in survival were not universally seen.3
- For shockable rhythms, trial protocols have directed that epinephrine be given after the third shock. The literature supports prioritizing defibrillation and CPR initially and giving epinephrine if initial attempts with CPR and defibrillation are not successful.3
- The recent systematic review3 found no difference in outcomes in trials comparing vasopressin alone or vasopressin combined with epinephrine to epinephrine alone for cardiac arrest, although these studies were underpowered.
- Multiple RCTs have compared high-dose with standard-dose epinephrine, and although some have shown higher rates of ROSC with high-dose epinephrine, none have shown improvement in survival to discharge or any longer-term outcomes.4–11
These recommendations are supported by the “2019 AHA Focused Update on Advanced Cardiovascular Life Support: Use of Advanced Airways, Vasopressors, and Extracorporeal CPR During Cardiac Arrest: An Update to the AHA Guidelines for CPR and Emergency Cardiovascular Care.”12
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-R | 1. Amiodarone or lidocaine may be considered for VF/pVT that is unresponsive to defibrillation. |
| 2b | C-LD | 2. For patients with OHCA, use of steroids during CPR is of uncertain benefit. |
| 3: No Benefit | B-NR | 3. Routine administration of calcium for treatment of cardiac arrest is not recommended. |
| 3: No Benefit | B-R | 4. Routine use of sodium bicarbonate is not recommended for patients in cardiac arrest. |
| 3: No Benefit | B-R | 5. The routine use of magnesium for cardiac arrest is not recommended. |
Synopsis
Pharmacological treatment of cardiac arrest is typically deployed when CPR with or without attempted defibrillation fails to achieve ROSC. This may include vasopressor agents such as epinephrine (discussed in Vasopressor Medications During Cardiac Arrest) as well as drugs without direct hemodynamic effects (“nonpressors”) such as antiarrhythmic medications, magnesium, sodium bicarbonate, calcium, or steroids (discussed here). Although theoretically attractive and of some proven benefit in animal studies, none of the latter therapies has been definitively proved to improve overall survival after cardiac arrest, although some may have possible benefit in selected populations and/or special circumstances.
Recommendations for the treatment of cardiac arrest due to hyperkalemia, including the use of calcium and sodium bicarbonate, are presented in Electrolyte Abnormalities. Recommendations for management of torsades de pointes are also presented in Torsades de Pointes.
Recommendation-Specific Supportive Text
- Administration of amiodarone or lidocaine to patients with OHCA was last formally reviewed in 20181 and demonstrated improved survival to hospital admission but did not improve overall survival to hospital discharge or survival with good neurological outcome.1,2 However, amiodarone and lidocaine each significantly improved survival to hospital discharge in a prespecified subgroup of patients with bystander-witnessed arrest, potentially arguing for a time-dependent benefit and a group for whom these drugs may be more useful. Other antiarrhythmic agents were not specifically addressed in the most recent evidence review and merit further evaluation. These include bretylium tosylate, which was recently reintroduced in the United States for treatment of immediately life-threatening ventricular arrhythmias but without any new information on its effectiveness or safety.3 Sotalol requires administration as a slow infusion, rendering it impractical to use in cardiac arrest.4 Similar limitations also apply to procainamide, although it has been given by rapid infusion as a second-line agent in cardiac arrest, with uncertain benefit.5 The efficacy of antiarrhythmic drugs when given in combination for cardiac arrest has not been systematically addressed and remains a knowledge gap. The role of prophylactic antiarrhythmic medications on ROSC after successful defibrillation is also uncertain. Though not associated with improved survival to hospital discharge, lidocaine decreased the recurrence of VF/pVT when administered prophylactically after successful defibrillation and ROSC.6 The “2018 AHA Focused Update on Advanced Cardiovascular Life Support Use of Antiarrhythmic Drugs During and Immediately After Cardiac Arrest: An Update to the AHA Guidelines for CPR and Emergency Cardiovascular Care”1 concluded that lidocaine use could be considered in specific circumstances (such as during EMS transport) when treatment of recurrent VF/pVT might be compromised. There is no evidence addressing the use of other antiarrhythmic drugs for this specific indication.
- Two randomized trials from the same center reported improved survival and neurological outcome when steroids were bundled in combination with vasopressin and epinephrine during cardiac arrest and also administered after successful resuscitation from cardiac arrest.7,8 However, nonrandomized studies of strictly intraarrest corticosteroid administration, in addition to standard resuscitation, show mixed outcomes.9,10 Due to the only studies suggesting benefit being from a single center with a bundled intervention, and observational data having conflicting results, whether steroids are beneficial during cardiac arrest remains unclear. At least 1 trial attempting to validate the findings of Mentzelopoulos et al is ongoing (NCT03640949).
- Since last addressed by the 2010 Guidelines, a 2013 systematic review found little evidence to support the routine use of calcium in undifferentiated cardiac arrest, though the evidence is very weak due calcium as a “last resort” medication in refractory cardiac arrest.11 Administration of calcium in special circumstances such as hyperkalemia and calcium blocker overdose is addressed in Electrolyte Abnormalities and Toxicity: β-Adrenergic Blockers and Calcium Channel Blockers.
- Clinical trials and observational studies since the 2010 Guidelines have yielded no new evidence that routine administration of sodium bicarbonate improves outcomes from undifferentiated cardiac arrest and evidence suggests that it may worsen survival and neurological recovery.12–14 Use of sodium bicarbonate in special circumstances such as hyperkalemia and drug overdose is addressed in Electrolyte Abnormalities and Toxicity: Sodium Channel Blockers, Including Tricyclic Antidepressants
- Magnesium’s role as an antiarrhythmic agent was last addressed by the 2018 focused update on advanced cardiovascular life support (ACLS) guidelines.1 RCTs have not found it to improve ROSC, survival or neurological outcome regardless of the presenting cardiac arrest rhythm,15–18 nor useful for monomorphic VT.19 There are anecdotal reports and small case series attesting to magnesium’s efficacy in the treatment of torsades de pointes (See Torsades de Pointes).
Recommendations 1 and 5 are supported by the 2018 focused update on ACLS guidelines.1 Recommendation 2 last received formal evidence review in 2015.20 Recommendations 3 and 4 last received formal evidence review in 2010.21
| COR | LOE | Recommendations |
|---|---|---|
| 2b | C-LD | 1. If an experienced sonographer is present and use of ultrasound does not interfere with the standard cardiac arrest treatment protocol, then ultrasound may be considered as an adjunct to standard patient evaluation, although its usefulness has not been well established. |
| 2b | C-LD | 2. When supplemental oxygen is available, it may be reasonable to use the maximal feasible inspired oxygen concentration during CPR. |
| 2b | C-LD | 3. An abrupt increase in end-tidal CO2 may be used to detect ROSC during compressions or when a rhythm check reveals an organized rhythm. |
| 2b | C-EO | 4. Routine measurement of arterial blood gases during CPR has uncertain value. |
| 2b | C-EO | 5. Arterial pressure monitoring by arterial line may be used to detect ROSC during chest compressions or when a rhythm check reveals an organized rhythm. |
Synopsis
Although the vast majority of cardiac arrest trials have been conducted in OHCA, IHCA comprises almost half of the arrests that occur in the United States annually, and many OHCA resuscitations continue into the emergency department. IHCA patients often have invasive monitoring devices in place such as central venous or arterial lines, and personnel to perform advanced procedures such as arterial blood gas analysis or point-of-care ultrasound are often present. Advanced monitoring such as ETCO2 monitoring is being increasingly used. Determining the utility of such physiological monitoring or diagnostic procedures is important. High-quality CPR, defibrillation when appropriate, vasopressors and/or antiarrhythmics, and airway management remain the cornerstones of cardiac arrest resuscitation, but some emerging data suggest that incorporating patient-specific imaging and physiological data into our approach to resuscitation holds some promise. See Metrics for High-Quality CPR for recommendations on physiological monitoring during CPR. More research in this area is clearly needed.
Recommendation-Specific Supportive Text
- Point-of-care cardiac ultrasound can identify cardiac tamponade or other potentially reversible causes of cardiac arrest and identify cardiac motion in pulseless electrical activity.1,2 However, cardiac ultrasound is also associated with longer interruptions in chest compressions.3 A single small RCT found no improvement in outcomes with the use of cardiac ultrasound during CPR.4
- No adult human studies directly compare levels of inspired oxygen concentration during CPR. A small number of studies has shown that higher Pao2 during CPR is associated with ROSC, but this is likely due to differences in patients or resuscitation quality.5–7
- Observational studies have found that increases in ETCO2 of more than 10 mm Hg may indicate ROSC, although no specific cutoff value indicative of ROSC has been identified.8
- Arterial Po2 and Pco2 values are dependent on cardiac output and ventilation and therefore will depend on both patient characteristics and CPR quality. One small study found wide discrepancies in blood gases between mixed venous and arterial samples during CPR and concluded that arterial samples are not accurate during resuscitation.9
- If an arterial line is in place, an abrupt increase in diastolic pressure or the presence of an arterial waveform during a rhythm check showing an organized rhythm may indicate ROSC.
Recommendations 1, 3, and 5 last received formal evidence review in 2015.10 Recommendation 2 last received formal evidence review in 2015,10 with an evidence update completed in 2020.11 Recommendation 4 last received formal evidence review in 2010.12
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. If termination of resuscitation (TOR) is being considered, BLS EMS providers should use the BLS termination of resuscitation rule where ALS is not available or may be significantly delayed. |
| 2a | B-NR | 2. It is reasonable for prehospital ALS providers to use the adult ALS TOR rule to terminate resuscitation efforts in the field for adult victims of OHCA. |
| 2a | B-NR | 3. In a tiered ALS- and BLS-provider system, the use of the BLS TOR rule can avoid confusion at the scene of a cardiac arrest without compromising diagnostic accuracy. |
| 2b | C-LD | 4. In intubated patients, failure to achieve an end-tidal CO2 of greater than 10 mm Hg by waveform capnography after 20 min of ALS resuscitation may be considered as a component of a multimodal approach to decide when to end resuscitative efforts, but it should not be used in isolation. |
| 3: No Benefit | C-LD | 5. We suggest against the use of point-of-care ultrasound for prognostication during CPR. |
| 3: Harm | C-EO | 6. In nonintubated patients, a specific end-tidal CO2 cutoff value at any time during CPR should not be used as an indication to end resuscitative efforts. |
Synopsis
OHCA is a resource-intensive condition most often associated with low rates of survival. It is important for EMS providers to be able to differentiate patients in whom continued resuscitation is futile from patients with a chance of survival who should receive continued resuscitation and transportation to hospital. This will aid in both resource utilization and optimizing a patient’s chance for survival. Using a validated TOR rule will help ensure accuracy in determining futile patients (Figures 5 and 6). Futility is often defined as less than 1% chance of survival,1 suggesting that for a TOR rule to be valid it should demonstrate high accuracy for predicting futility with the lower confidence limit greater than 99% on external validation.
Recommendation-Specific Supportive Text
- The BLS TOR rule recommends TOR when all of the following criteria apply before moving to the ambulance for transport: (1) arrest was not witnessed by EMS providers or first responder; (2) no ROSC obtained; and (3) no shocks were delivered. In a recent meta-analysis of 7 published studies (33 795 patients), only 0.13% (95% CI, 0.03%– 0.58%) of patients who fulfilled the BLS termination criteria survived to hospital discharge.3
- The ALS TOR rule recommends TOR when all of the following criteria apply before moving to the ambulance for transport: (1) arrest was not witnessed; (2) no bystander CPR was provided; (3) no ROSC after full ALS care in the field; and (4) no AED shocks were delivered. In a recent meta-analysis of 2 published studies (10 178 patients), only 0.01% (95% CI, 0.00%–0.07%) of patients who fulfilled the ALS termination criteria survived to hospital discharge.3
- The BLS TOR rule, otherwise known as the universal TOR rule (arrest not witnessed by EMS providers; no shock delivered; no ROSC), has been prospectively validated in combined BLS and ALS systems.4 Although the rule did not have adequate specificity after 6 minutes of resuscitation (false-positive rate: 2.1%) it did achieve better than 99% specificity after approximately 15 minutes of attempted resuscitation, while still reducing transportation by half. A retrospective analysis found that application of the universal TOR at 20 minutes of resuscitation was able to predict futility, identifying over 99% of survivors and patients with good neurological outcome.5
- In intubated patients, an ETCO2 measurement less than 10 mm Hg indicates low to no blood flow. Several small studies provide evidence showing that an ETCO2 less than 10 mm Hg after 20 minutes of ALS resuscitation is strongly but not perfectly predictive of futility.6–9 These small observational studies suffer from high risk of bias. Alternative ETCO2 thresholds and timepoints have been proposed. The use of ETCO2 alone to predict patient outcome needs to be validated in a large prospective study.
- A recent systematic review found that no sonographic finding had consistently high sensitivity for clinical outcomes to be used as the sole criterion to terminate cardiac arrest resuscitation.10 Although some findings demonstrated higher ranges of sensitivity and/or specificity, studies examining the use of point-of-care ultrasound during cardiac arrest demonstrate varying results and are hindered by significant bias. There is considerable heterogeneity between studies in terms of timing and application of point-of-care ultrasound as well as inconsistent definitions and terminology in terms of cardiac motion. Further there is little research examining the interrater reliability of ultrasound findings during cardiac arrest.11,12 In addition, see Adjuncts to CPR for ultrasound as an adjunct to CPR.
- No studies were found that specifically examined the use of ETCO2 in cardiac arrest patients without an advanced airway. It is not known whether ETCO2 values during bag-mask ventilation are as reliable as those with an advanced airway in place. Because of the lack of evidence, there is nothing to support using any cutoff value of ETCO2 for decisions about TOR in a nonintubated patient.
Recommendations 1, 2, 3, and 5 are supported by the 2020 CoSTRs for BLS and ALS.13,14 Recommendations 4 and 6 last received formal evidence review in 2015.15
AED indicates automated external defibrillator; and BLS, basic life support.
ACLS indicates advanced cardiovascular life support; and CPR, cardiopulmonary resuscitation.
Introduction
Airway management during cardiac arrest usually commences with a basic strategy such as bag-mask ventilation. In addition, it may be helpful for providers to master an advanced airway strategy as well as a second (backup) strategy for use if they are unable to establish the first-choice airway adjunct. Because placement of an advanced airway may result in interruption of chest compressions, a malpositioned device, or undesirable hyperventilation, providers should carefully weigh these risks against the potential benefits of an advanced airway. The 2019 focused update on ACLS guidelines addressed the use of advanced airways in cardiac arrest and noted that either bag-mask ventilation or an advanced airway strategy may be considered during CPR for adult cardiac arrest in any setting.1 Outcomes from advanced airway and bag-mask ventilation interventions are highly dependent on the skill set and experience of the provider (Figure 7). Thus, the ultimate decision of the use, type, and timing of an advanced airway will require consideration of a host of patient and provider characteristics that are not easily defined in a global recommendation. Important considerations for determining airway management strategies is provider airway management skill and experience, frequent retraining for providers, and ongoing quality improvement to minimize airway management complications.
| COR | LOE | Recommendation |
|---|---|---|
| 2b | B-R | 1. Either bag-mask ventilation or an advanced airway strategy may be considered during CPR for adult cardiac arrest in any setting depending on the situation and skill set of the provider. |
Recommendation-Specific Supportive Text
- One large RCT in OHCA comparing bag-mask ventilation with endotracheal intubation (ETI) in a physician-based EMS system showed no significant benefit for either technique for 28-day survival or survival with favorable neurological outcome.2 The success rate of ETI in this study was 98%, suggesting a relatively optimal setting for the potential success of ETI as an intervention. Further research is required to determine equivalence or superiority between the 2 approaches for acute airway management.
These recommendations are supported by the 2019 focused update on ACLS guidelines.1
| COR | LOE | Recommendations |
|---|---|---|
| 2a | B-R | 1. If an advanced airway is used, a supraglottic airway can be used for adults with OHCA in settings with low tracheal intubation success rates or minimal training opportunities for endotracheal tube placement. |
| 2a | B-R | 2. If an advanced airway is used, either a supraglottic airway or endotracheal intubation can be used for adults with OHCA in settings with high tracheal intubation success rates or optimal training opportunities for endotracheal tube placement. |
| 2a | B-R | 3. If an advanced airway is used in the in-hospital setting by expert providers trained in these procedures, either a supraglottic airway or an endotracheal tube placement can be used. |
Recommendation-Specific Supportive Text
- 2, and 3. One RCT in OHCA comparing SGA (with iGel) to ETI in a non–physician-based EMS system (ETI success, 69%) found no difference in survival or survival with favorable neurological outcome at hospital discharge.3 A second RCT in OHCA comparing SGA (with laryngeal tube) with ETI in a non–physician-based EMS system (ETI success, 52%) found both better survival to hospital discharge and better survival to hospital discharge with good neurological outcome in the patients managed with SGA.4 These results are challenging to contextualize because they both allowed for provider deviation from protocol based on clinical judgment. Additionally, precise thresholds for high or low tracheal intubation success rates have not been identified, though guidance can be taken from the existing clinical trials. Thus, it is difficult to understand the potential benefit (or harm), per individual, that drove the decision to place the specific advanced airway device. The decision on placement of an advanced airway requires an understanding of patient and provider characteristics that are not easily defined in a global recommendation. Because of a paucity of studies on advanced airway management for IHCA, the IHCA recommendations are extrapolated from OHCA data. Based on these issues, there is a need for further research specifically on the interface between patient factors and the experience, training, tools, and skills of the provider. Given these reasons, a recommendation for SGA in preference to ETI would be premature.
These recommendations are supported by the 2019 focused update on ACLS guidelines.1
| 1 | B-NR | 1. Frequent experience or frequent retraining is recommended for providers who perform endotracheal intubation. |
|---|---|---|
| 1 | C-LD | 2. If advanced airway placement will interrupt chest compressions, providers may consider deferring insertion of the airway until the patient fails to respond to initial CPR and defibrillation attempts or obtains ROSC. |
| 1 | C-LD | 3. Continuous waveform capnography is recommended in addition to clinical assessment as the most reliable method of confirming and monitoring correct placement of an endotracheal tube. |
| 1 | C-EO | 4. EMS systems that perform prehospital intubation should provide a program of ongoing quality improvement to minimize complications and track overall supraglottic airway and endotracheal tube placement success rates. |
Recommendation-Specific Supportive Text
- To maintain provider skills from initial training, frequent retraining is important.5,6 However, future research will need to address the specific type, amount, and duration between training experiences.
- Although an advanced airway can be placed without interrupting chest compressions,7 unfortunately, such interruptions still occur. Therefore, providers should weigh the potential benefits of an advanced airway with the benefits of maintaining a high chest compression fraction.8–10
- In a small clinical trial and several observational studies, waveform capnography was 100% specific for confirming endotracheal tube position during cardiac arrest.11–13 The sensitivity of waveform capnography decreases after a prolonged cardiac arrest.11–13 The use of waveform capnography to assess the placement of other advanced airways (eg, Combitube, laryngeal mask airway) has not been studied.
- The rationale for tracking the overall success rate for systems performing ETI is to make informed decisions as to whether practice should allow for ETI, move toward SGA, or simply use bag-mask ventilation for patients in cardiac arrest; recommendations will vary depending on the overall success rate in a given system.
These recommendations are supported by the 2019 focused update on ACLS guidelines.1
Introduction
Many alternatives and adjuncts to conventional CPR have been developed. These include mechanical CPR, impedance threshold devices (ITD), active compression-decompression (ACD) CPR, and interposed abdominal compression CPR. Many of these techniques and devices require specialized equipment and training.
Mechanical CPR devices deliver automated chest compressions, thereby eliminating the need for manual chest compressions. There are 2 different types of mechanical CPR devices: a load-distributing compression band that compresses the entire thorax circumferentially and a pneumatic piston device that compresses the chest in an anteroposterior direction. A recent systematic review of 11 RCTs (overall moderate to low certainty of evidence) found no evidence of improved survival with good neurological outcome with mechanical CPR compared with manual CPR in either OHCA or IHCA.1 Given the perceived logistic advantages related to limited personnel and safety during patient transport, mechanical CPR remains popular among some providers and systems.
ACD-CPR is performed by using a handheld device with a suction cup applied to the midsternum, actively lifting up the chest during decompressions, thereby enhancing the negative intrathoracic pressure generated by chest recoil and increasing venous return and cardiac output during the next chest compression. The ITD is a pressure-sensitive valve attached to an advanced airway or face mask that limits air entry into the lungs during the decompression phase of CPR, enhancing the negative intrathoracic pressure generated during chest wall recoil and improving venous return and cardiac output during CPR.
There are many alternative CPR techniques being used, and many are unproven. As an example, there is insufficient evidence concerning the cardiac arrest bundle of care with the inclusion of “heads-up” CPR to provide a recommendation concerning its use.2 Further investigation in this and other alternative CPR techniques is best explored in the context of formal controlled clinical research
| COR | LOE | Recommendations |
|---|---|---|
| 2b | C-LD | 1. The use of mechanical CPR devices may be considered in specific settings where the delivery of high-quality manual compressions may be challenging or dangerous for the provider, as long as rescuers strictly limit interruptions in CPR during deployment and removal of the device. |
| 3: No Benefit | B-R | 2. The routine use of mechanical CPR devices is not recommended. |
Recommendation-Specific Supportive Text
- and 2. Studies of mechanical CPR devices have not demonstrated a benefit when compared with manual CPR, with a suggestion of worse neurological outcome in some studies. In the ASPIRE trial (1071 patients), use of the load-distributing band device was associated with similar odds of survival to hospital discharge (adjusted odds ratio [aOR], 0.56; CI, 0.31–1.00; P=0.06), and worse survival with good neurological outcome (3.1% versus 7.5%; P=0.006), compared with manual CPR.3 In the CIRC trial (n=4231), use of load-distributing band–CPR resulted in statistically equivalent rates of survival to hospital discharge (aOR, 1.06; CI, 0.83–1.37) and survival with good neurological outcome (aOR, 0.80; CI, 0.47–1.37).4 In the PARAMEDIC trial (n=4470), use of a mechanical piston device produced similar rates of 30-day survival (aOR, 0.86; CI, 0.64–1.15), and worse survival with good neurological outcome (aOR, 0.72; CI, 0.52–0.99), compared with manual CPR.5 In the LINC trial (n=2589), survival with good neurological outcome was similar in both groups (8.3% versus 7.8%; risk difference, 0.55%; 95% CI, –1.5% to 2.6%).6
Acknowledging these data, the use of mechanical CPR devices by trained personnel may be beneficial in settings where reliable, high-quality manual compressions are not possible or may cause risk to personnel (ie, limited personnel, moving ambulance, angiography suite, prolonged resuscitation, or with concerns for infectious disease exposure).
This topic last received formal evidence review in 2015.7
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. The effectiveness of active compression-decompression CPR is uncertain. Active compression-decompression CPR might be considered for use when providers are adequately trained and monitored. |
| 2b | C-LD | 2. The combination of active compression-decompression CPR and impedance threshold device may be reasonable in settings with available equipment and properly trained personnel. |
| 3: No Benefit | A | 3. The routine use of the impedance threshold device as an adjunct during conventional CPR is not recommended. |
Recommendation-Specific Supportive Text
- A 2013 Cochrane review of 10 trials comparing ACD-CPR with standard CPR found no differences in mortality and neurological function in adults with OHCA or IHCA.8 An important added consideration with this modality is that of increased rescuer fatigue, which could impair the overall quality of CPR.
- ACD-CPR and ITD may act synergistically to enhance venous return during chest decompression and improve blood flow to vital organs during CPR. The ResQTrial demonstrated that ACD plus ITD was associated with improved survival to hospital discharge with favorable neurological function for OHCA compared with standard CPR, though this study was limited by a lack of blinding, different CPR feedback elements between the study arms (ie, cointervention), lack of CPR quality assessment, and early TOR.9,10 The 2015 AHA Guidelines Update for CPR and Emergency Cardiovascular Care7 evaluated this topic and noted that though a large RCT of low-quality demonstrated benefit of its use, additional trials were needed to confirm the results because of study limitations noted. Thus, ACD-CPR plus ITD was not recommended in previous versions of the AHA Guidelines. However, in settings where the equipment and trained personnel are available, ACD-CPR plus ITD could be an alternative to standard CPR.
- In the PRIMED study (n=8178), the use of the ITD (compared with a sham device) did not significantly improve survival to hospital discharge or survival with good neurological function in patients with OHCA.11 Despite the addition of a post hoc analysis of the PRIMED trial for ITD,12 the routine use of the ITD as an adjunct during conventional CPR is not recommended.
This topic last received formal evidence review in 2015.7
| COR | LOE | Recommendation |
|---|---|---|
| 2b | B-NR | 1. Interposed abdominal compression CPR may be considered during in-hospital resuscitation when sufficient personnel trained in its use are available. |
Recommendation-Specific Supportive Text
- Interposed abdominal compression CPR is a 3-rescuer technique that includes conventional chest compressions combined with alternating abdominal compressions. The dedicated rescuer who provides manual abdominal compressions will compress the abdomen midway between the xiphoid and the umbilicus during the relaxation phase of chest compression. This topic was last reviewed in 2010 and identified 2 randomized trials, interposed abdominal compression CPR performed by trained rescuers improved short-term survival13 and survival to hospital discharge,14 compared with conventional CPR for adult IHCA. One RCT of adult OHCA15 did not show any survival advantage to interposed abdominal compression CPR. More evaluation is needed to further define the routine use of this technique.
This topic last received formal evidence review in 2010.16
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. There is insufficient evidence to recommend the routine use of extracorporeal CPR (ECPR) for patients with cardiac arrest. ECPR may be considered for select cardiac arrest patients for whom the suspected cause of the cardiac arrest is potentially reversible during a limited period of mechanical cardiorespiratory support. |
Synopsis
ECPR refers to the initiation of cardiopulmonary bypass during the resuscitation of a patient in cardiac arrest. This involves the cannulation of a large vein and artery and initiation of venoarterial extracorporeal circulation and membrane oxygenation (ECMO) (Figure 8). The goal of ECPR is to support end organ perfusion while potentially reversible conditions are addressed. ECPR is a complex intervention that requires a highly trained team, specialized equipment, and multidisciplinary support within a healthcare system. The 2019 focused update on ACLS guidelines1 addressed the use of ECPR for cardiac arrest and noted that there is insufficient evidence to recommend the routine use of ECPR in cardiac arrest. However, ECPR may be considered if there is a potentially reversible cause of an arrest that would benefit from temporary cardiorespiratory support. One important consideration is the selection of patients for ECPR and further research is needed to define patients who would most benefit from the intervention. Furthermore, the resource intensity required to begin and maintain an ECPR program should be considered in the context of strengthening other links in the Chain of Survival. Additional investigations are necessary to evaluate cost-effectiveness, resource allocation, and ethics surrounding the routine use of ECPR in resuscitation.
Recommendation-Specific Supportive Text
- There are no RCTs on the use of ECPR for OHCA or IHCA. Fifteen observational studies were identified for OHCA that varied in inclusion criteria, ECPR settings, and study design, with the majority of studies reporting improved neurological outcome associated with ECPR.2 For ECPR use in the in-hospital setting, all studies were assessed as having very serious risk of bias (primarily due to confounding) and the overall certainty of evidence was rated as very low for all outcomes.2 In 3 studies, ECPR was not associated with beneficial effects for short- or long-term neurological outcomes,3–5 while 1 study6 did report associated short- and long-term neurological outcome benefit. Despite many studies reporting favorable outcomes with the use of ECPR, the vast majority of the studies are from single centers with varying inclusion criteria and settings, with decisions to perform ECPR made on a case-by-case basis. While there is currently no evidence to clearly define what should constitute “selected patients,” most of the studies analyzed included younger patients with fewer comorbidities. More data are clearly needed from studies of higher methodologic quality, including randomized trials.
These recommendations are supported by the 2019 focused update on ACLS guidelines.1
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. In hemodynamically stable patients, IV adenosine may be considered for treatment and aiding rhythm diagnosis when the cause of the regular, monomorphic rhythm cannot be determined. |
| 2b | B-R | 2. Administration of IV amiodarone, procainamide, or sotalol may be considered for the treatment of wide-complex tachycardia. |
| 3: Harm | B-NR | 3. Verapamil should not be administered for any wide-complex tachycardia unless known to be of supraventricular origin and not being conducted by an accessory pathway. |
| 3: Harm | C-LD | 4. Adenosine should not be administered for hemodynamically unstable, irregularly irregular, or polymorphic wide-complex tachycardias. |
Synopsis
A wide-complex tachycardia is defined as a rapid rhythm (generally 150 beats/min or more when attributable to an arrhythmia) with a QRS duration of 0.12 seconds or more. It can represent any aberrantly conducted supraventricular tachycardia (SVT), including paroxysmal SVT caused by atrioventricular (AV) reentry, aberrantly conducted atrial fibrillation, atrial flutter, or ectopic atrial tachycardia. A wide-complex tachycardia can also be caused by any of these supraventricular arrhythmias when conducted by an accessory pathway (called pre-excited arrhythmias). Conversely, a wide-complex tachycardia can also be due to VT or a rapid ventricular paced rhythm in patients with a pacemaker.
Initial management of wide-complex tachycardia requires a rapid assessment of the patient’s hemodynamic stability. Unstable patients require immediate electric cardioversion. If hemodynamically stable, a presumptive rhythm diagnosis should be attempted by obtaining a 12-lead ECG to evaluate the tachycardia’s features. This includes identifying P waves and their relationship to QRS complexes and (in the case of patients with a pacemaker) pacing spikes preceding QRS complexes.
A wide-complex tachycardia can be regular or irregularly irregular and have uniform (monomorphic) or differing (polymorphic) QRS complexes from beat to beat. Each of these features can also be useful in making a presumptive rhythm diagnosis. An irregularly irregular wide-complex tachycardia with monomorphic QRS complexes suggests atrial fibrillation with aberrancy, whereas pre-excited atrial fibrillation or polymorphic VT are likely when QRS complexes change in their configuration from beat to beat. Conversely, a regular wide-complex tachycardia could represent monomorphic VT or an aberrantly conducted reentrant paroxysmal SVT, ectopic atrial tachycardia, or atrial flutter. Distinguishing between these rhythm etiologies is the key to proper drug selection for treatment. While hemodynamically stable rhythms afford an opportunity for evaluation and pharmacological treatment, the need for prompt electric cardioversion should be anticipated in the event the arrhythmia proves unresponsive to these measures or rapid decompensation occurs. A more detailed approach to rhythm management is found elsewhere.1–3
Recommendation-Specific Supportive Text
- Before embarking on empirical drug therapy, obtaining a 12-lead ECG and/or seeking expert consultation for diagnosis is encouraged, if available. If a regular wide-complex tachycardia is suspected to be paroxysmal SVT, vagal maneuvers can be considered before initiating pharmacological therapies (see Regular Narrow-Complex Tachycardia). Adenosine is an ultra–short-acting drug that is effective in terminating regular tachycardias when caused by AV reentry. Adenosine will not typically terminate atrial arrhythmias (such as atrial flutter or atrial tachycardia) but will transiently slow the ventricular rate by blocking conduction of P waves through the AV node, afford their recognition, and help establish the rhythm diagnosis. While ineffective in terminating ventricular arrhythmias, adenosine’s relatively short-lived effect on blood pressure makes it less likely to destabilize monomorphic VT in an otherwise hemodynamically stable patient. These features make adenosine relatively safe for treating a hemodynamically stable, regular, monomorphic wide-complex tachycardia of unknown type4 and as an aid in rhythm diagnosis, although its use is not completely without risk.5,6
- IV antiarrhythmic medications may be considered in stable patients with wide-complex tachycardia, particularly if suspected to be VT or having failed adenosine. Because of their longer duration of action, antiarrhythmic agents may also be useful to prevent recurrences of wide-complex tachycardia. Lidocaine is not included as a treatment option for undifferentiated wide-complex tachycardia because it is a relatively “narrow-spectrum” drug that is ineffective for SVT, probably because its kinetic properties are less effective for VT at hemodynamically tolerated rates than amiodarone, procainamide, or sotalol are.7–10 In contrast, amiodarone, procainamide, and sotalol are “broader-spectrum” antiarrhythmics than lidocaine and can treat both SVT and VT, but they can cause hypotension. Since the 2010 Guidelines, a new branded bioequivalent formulation of amiodarone has become available for IV infusion with less hypotensive effects than the older generic formulation.11 There are few direct comparisons of efficacy between amiodarone, procainamide, and sotalol themselves,12 which the writing group felt were insufficient to favor one of these drugs over another, apart from cautioning about their use in patients with long QT, amiodarone in suspected pre-excited arrhythmias, or giving these drugs in combination without prior expert consultation. Any of these drugs can also worsen wide-complex tachycardia, converting it to an arrhythmia that is more rapid, less hemodynamically stable, or more malignant, such that availability of a defibrillator is encouraged when these drugs are administered.13
- Verapamil is a calcium channel blocking agent that slows AV node conduction, shortens the refractory period of accessory pathways, and acts as a negative inotrope and vasodilator. Its effects are mediated by a different mechanism and are longer lasting than adenosine. Though effective for treating a wide-complex tachycardia known to be of supraventricular origin and not involving accessory pathway conduction, verapamil’s negative inotropic and hypotensive effects can destabilize VT14 and accelerate pre-excited atrial fibrillation and flutter.15 Similar concerns may also apply to other drugs commonly used to treat SVTs, such as diltiazem and β-adrenergic blockers, which are not addressed in this recommendation and require evidence review.
- The combination of adenosine’s short-lived slowing of AV node conduction, shortening of refractoriness in the myocardium and accessory pathways, and hypotensive effects make it unsuitable in hemodynamically unstable patients and for treating irregularly irregular and polymorphic wide-complex tachycardias. Adenosine only transiently slows irregularly irregular rhythms, such as atrial fibrillation, rendering it unsuitable for their management. The drug’s hypotensive and tissue refractoriness–shortening effects can accelerate ventricular rates in polymorphic VT and, when atrial fibrillation or flutter are conducted by an accessory pathway, risk degeneration to VF.16 Thus, the drug is not recommended in hemodynamically unstable patients or for treating irregularly irregular or polymorphic wide-complex tachycardias.
This topic last received formal evidence review in 2010.17
| COR | LOE | Recommendation |
|---|---|---|
| 2a | C-LD | 1. If pharmacological therapy is unsuccessful for the treatment of a hemodynamically stable wide-complex tachycardia, cardioversion or seeking urgent expert consultation is reasonable. |
Recommendation-Specific Supportive Text
- When available, expert consultation can be helpful to assist in the diagnosis and management of treatment-refractory wide-complex tachycardia. Electric cardioversion can be useful either as firstline treatment or for drug-refractory wide-complex tachycardia due to reentry rhythms (such as atrial fibrillation, atrial flutter, AV reentry, and VT). However, electric cardioversion may not be effective for automatic tachycardias (such as ectopic atrial tachycardias), entails risks associated with sedation, and does not prevent recurrences of the wide-complex tachycardia. Notably, when the QRS complex is of uniform morphology, shock synchronized to the QRS is encouraged because this minimizes the risk of provoking VF by a mistimed shock during the vulnerable period of the cardiac cycle (T wave).18 In contrast, polymorphic wide-complex tachycardias cannot be synchronized reliably because of the differing characteristics of each QRS complex, and require high-energy defibrillation.19
This topic last received formal evidence review in 2010.17
Synopsis
Polymorphic VT refers to a wide-complex tachycardia of ventricular origin with differing configurations of the QRS complex from beat to beat. However, the most critical feature in the diagnosis and treatment of polymorphic VT is not the morphology of rhythm but rather what is known (or suspected) about the patient’s underlying QT interval. Torsades de pointes is a form of polymorphic VT that is associated with a prolonged heart rate–corrected QT interval when the rhythm is normal and VT is not present. The risk for developing torsades increases when the corrected QT interval is greater than 500 milliseconds and accompanied by bradycardia.1 Torsades can be due to an inherited genetic abnormality2 and can also be caused by drugs and electrolyte imbalances that cause lengthening of the QT interval.3
Conversely, polymorphic VT not associated with a long QT is most often due to acute myocardial ischemia.4,5 Other potential causes include catecholaminergic polymorphic VT, a genetic abnormality in which polymorphic VT is provoked by exercise or emotion in the absence of QT prolongation6 ; “short QT” syndrome, a form of polymorphic VT associated with an unusually short QT interval (corrected QT interval less than 330–370 milliseconds)7,8 ; and bidirectional VT seen in digitalis toxicity in which the axis of alternate QRS complexes shifts by 180 degrees.9 Supportive data for the acute pharmacological treatment of polymorphic VT, with and without long corrected QT interval, is largely based on case reports and case series, because no RCTs exist.
| COR | LOE | Recommendation |
|---|---|---|
| 1 | B-NR | 1. Immediate defibrillation is recommended for sustained, hemodynamically unstable polymorphic VT. |
Recommendation-Specific Supportive Text
- Regardless of the underlying QT interval, all forms of polymorphic VT tend to be hemodynamically and electrically unstable. They may repeatedly recur and remit spontaneously, become sustained, or degenerate to VF, for which electric shock may be required. When the QRS complex of a VT is of uniform morphology, electric cardioversion with the shock synchronized to the QRS minimizes the risk of provoking VF by a mistimed shock during the vulnerable period of the cardiac cycle (T wave).10 In contrast, polymorphic VT cannot be synchronized reliably because of the differing characteristics of each QRS complex and requires high-energy unsynchronized defibrillation.11 While effective in terminating polymorphic VT, electric shock may not prevent its recurrence, for which pharmacological therapies are often required and the primary focus of the ensuing recommendations
This topic last received formal evidence review in 2010.12
| COR | LOE | Recommendation |
|---|---|---|
| 2b | C-LD | 1. Magnesium may be considered for treatment of polymorphic VT associated with a long QT interval (torsades de pointes). |
Recommendation-Specific Supportive Text
- Torsades de pointes typically presents in a recurring pattern of self-terminating, hemodynamically unstable polymorphic VT in context of a known or suspected long QT abnormality, often with an associated bradycardia. Immediate defibrillation is the treatment of choice when torsades is sustained or degenerates to VF. However, termination of torsades by shock does not prevent its recurrence, which requires additional measures. In small case series, IV magnesium has been effective in suppressing and preventing recurrences of torsades.13–16 Magnesium is believed to suppress early afterdepolarizations, which are fluctuations in the myocardial action potential that can trigger the salvos of VT seen in torsades.17 Correcting any electrolyte abnormalities, particularly hypokalemia, is also advisable. Torsades is not treatable with antiarrhythmic medications, which can themselves prolong the QT interval and promote the arrhythmia. When given acutely, β-adrenergic blockers can also precipitate torsades by causing or worsening bradycardia. In patients with bradycardia or pause-precipitated torsades, expert consultation is best sought for additional measures such as overdrive pacing or isoproterenol,18–20 if needed. The use of magnesium in torsades de pointes was addressed by the 2010 Guidelines and updated in a 2018 focused update on ACLS guidelines,21 with an interim evidence review that identified no new information that would modify previous recommendations.
This topic last received formal evidence review in 2010.12
| COR | LOE | Recommendations |
|---|---|---|
| 2b | C-LD | 1. IV lidocaine, amiodarone, and measures to treat myocardial ischemia may be considered to treat polymorphic VT in the absence of a prolonged QT interval. |
| 3: No Benefit | C-LD | 2. We do not recommend routine use of magnesium for the treatment of polymorphic VT with a normal QT interval. |
Recommendation-Specific Supportive Text
- Polymorphic VT that is not associated with QT prolongation is often triggered by acute myocardial ischemia and infarction,4,5 often rapidly degenerates into VF, and is treated similarly to other ventricular arrhythmias (VT and VF). However, termination of polymorphic VT with defibrillation may not prevent its recurrence, which often requires additional measures. No RCTs have been performed to determine the best practice for pharmacological management of polymorphic VT. However measures to treat myocardial ischemia (eg, β-adrenergic blockers or emergent coronary intervention) as well as lidocaine and amiodarone may be effective22–29 in concert with defibrillation when the arrhythmia is sustained. β-Adrenergic blockers have also been shown to reduce the incidence of ventricular arrhythmias in acute coronary syndromes.30,31 Expert consultation is advisable when other causes of polymorphic VT are suspected, for which β-adrenergic blockers and antiarrhythmics may also have efficacy.6,32 This topic was last addressed by the 2010 Guidelines, with an interim evidence update that identified no new information that would modify previous recommendations. Newer defined diagnostic entities causing polymorphic VT merit future evidence evaluation.
- In the absence of long QT, magnesium has not been shown to be effective in the treatment of polymorphic VT 13 or to afford benefit in the acute management of other ventricular tachyarrhythmias.16
These recommendations are supported by the 2018 focused update on ACLS guidelines.21
Introduction
Management of SVTs is the subject of a recent joint treatment guideline from the AHA, the American College of Cardiology, and the Heart Rhythm Society.1
Narrow-complex tachycardia represents a range of tachyarrhythmias originating from a circuit or focus involving the atria or the AV node. Clinicians must determine if the tachycardia is narrow-complex or wide-complex tachycardia and if it has a regular or irregular rhythm. For patients with a sinus tachycardia (heart rate greater than 100/min, P waves), no specific drug treatment is needed, and clinicians should focus on identification and treatment of the underlying cause of the tachycardia (fever, dehydration, pain). If the patient presents with SVT, the primary goal of treatment is to quickly identify and treat patients who are hemodynamically unstable (ischemic chest pain, altered mental status, shock, hypotension, acute heart failure) or symptomatic due to the arrhythmia. Synchronized cardioversion or drugs or both may be used to control unstable or symptomatic regular narrow-complex tachycardia. The available evidence suggests no appreciable differences in success or major adverse event rates between calcium channel blockers and adenosine.2
In patients with narrow-complex tachycardia who are refractory to the measures described, this may indicate a more complicated rhythm abnormality for which expert consultation may be advisable.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. Synchronized cardioversion is recommended for acute treatment in patients with hemodynamically unstable SVT. |
| 1 | B-NR | 2. Synchronized cardioversion is recommended for acute treatment in patients with hemodynamically stable SVT when vagal maneuvers and pharmacological therapy is ineffective or contraindicated. |
Recommendation-Specific Supportive Text
- and 2. Management of hemodynamically unstable patients with SVT must start with prompt restoration of sinus rhythm through the use of cardioversion. Cardioversion has been shown to be both safe and effective in the prehospital setting for hemodynamically unstable patients with SVT who had failed to respond to vagal maneuvers and IV pharmacological therapies.3 Cardioversion is advised in patients who present with hypotension, acutely altered mental status, signs of shock, chest pain, or acute heart failure. Though rare, cardioversion may also be necessary in stable patients with SVT. Most stable patients with SVT have high conversion success rates of 80% to 98% with pharmacological management (eg, adenosine, diltiazem).4,5 However, if drugs fail to restore sinus rhythm, cardioversion is safe and effective for stable patients after adequate sedation and anesthesia.
Adult Tachycardia with a Pulse Algorithm
In the absence of knowing the manufacturer’s recommendation for appropriate energy settings, the previous 2010 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care (and reaffirmed in 2015) recommendations for synchronized cardioversion are still applicable [Narrow regular: 50-100 J; Narrow irregular: 120-200 J biphasic or 200 J monophasic; Wide regular: 100 J; Wide irregular: defibrillation dose (not synchronized)].
These recommendations are supported by the “2015 ACC/AHA/HRS Guideline for the Management of Adult Patients With SVT: A Report of the American College of Cardiology/AHA Task Force on Clinical Practice Guidelines and the Heart Rhythm Society.”6
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-R | 1. Vagal maneuvers are recommended for acute treatment in patients with SVT at a regular rate. |
| 1 | B-R | 2. Adenosine is recommended for acute treatment in patients with SVT at a regular rate. |
| 2a | B-R | 3. IV diltiazem or verapamil can be effective for acute treatment in patients with hemodynamically stable SVT at a regular rate. |
| 2a | C-LD | 4. IV β-adrenergic blockers are reasonable for acute treatment in patients with hemodynamically stable SVT at a regular rate. |
Recommendation-Specific Supportive Text
- Success rates for the Valsalva maneuver in terminating SVT range from 19% to 54%.7 Augmenting the Valsalva maneuver with passive leg raise is more effective.8 Caution is advised when deploying carotid massage in older patients given the potential thromboembolic risk.
- The 2015 American College of Cardiology, AHA, and Heart Rhythm Society Guidelines evaluated and recommended adenosine as a first-line treatment for regular SVT because of its effectiveness, extremely short half-life, and favorable side-effect profile.6 A Cochrane systematic review of 7 RCTs (622 patients) found similar rates of conversion to sinus rhythm with adenosine or calcium channel blockers (90% versus 93%) and no significant difference in hypotension.2 Adenosine may have profound effects in post–heart transplant patients and can cause severe bronchospasm in asthma patients.
- Treatment of hemodynamically stable patients with IV diltiazem or verapamil have been shown to convert SVT to normal sinus rhythm in 64% to 98% of patients.4,9–11 These agents are particularly useful in patients who cannot tolerate β-adrenergic blockers or who have recurrent SVT after treatment with adenosine. Caution should be taken to administer these medications slowly to decrease the potential for hypotension.11 Diltiazem and verapamil are not appropriate in the setting of suspected systolic heart failure.6
- Evidence for the effectiveness of β-adrenergic blockers in terminating SVT is limited. In a trial that compared esmolol with diltiazem, diltiazem was more effective in terminating SVT.5 Nonetheless, β-adrenergic blockers are generally safe, and it is reasonable to use them to terminate SVT in hemodynamically stable patients.6
These recommendations are supported by the 2015 American College of Cardiology, AHA, and Heart Rhythm Society Guidelines for the Management of Adult Patients With SVT.6
Introduction
Atrial fibrillation is an SVT consisting of disorganized atrial electric activation and uncoordinated atrial contraction. Atrial flutter is an SVT with a macroreentrant circuit resulting in rapid atrial activation but intermittent ventricular response. These arrhythmias are common and often coexist, and their treatment recommendations are similar.
Treatment of atrial fibrillation/flutter depends on the hemodynamic stability of the patient as well as prior history of arrhythmia, comorbidities, and responsiveness to medication. Hemodynamically unstable patients and those with rate-related ischemia should receive urgent electric cardioversion. Hemodynamically stable patients can be treated with a rate-control or rhythm-control strategy. Rate control is more common in the emergency setting, using IV administration of a nondihydropyridine calcium channel antagonist (eg, diltiazem, verapamil) or a β-adrenergic blocker (eg, metoprolol, esmolol). While amiodarone is typically considered a rhythm-control agent, it can effectively reduce ventricular rate with potential use in patients with congestive heart failure where β-adrenergic blockers may not be tolerated and nondihydropyridine calcium channel antagonists are contraindicated. Long-term anticoagulation may be necessary for patients at risk for thromboembolic events based on their CHA2 DS2 - VASc score. The choice of anticoagulation is beyond the scope of these guidelines.
The rhythm-control strategy (sometimes called chemical cardioversion) includes antiarrhythmic medications given to convert the rhythm to sinus and/or prevent recurrent atrial fibrillation/flutter (Table 3). Patient selection, evaluation, timing, drug selection, and anticoagulation for patients undergoing rhythm control are beyond the scope of these guidelines and are presented elsewhere.1,2
The management of patients with preexcitation syndromes (aka Wolff-Parkinson-White) is covered in the Wide-Complex Tachycardia section.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Hemodynamically unstable patients with atrial fibrillation or atrial flutter with rapid ventricular response should receive electric cardioversion. |
| 1 | C-LD | 2. Urgent direct-current cardioversion of new-onset atrial fibrillation in the setting of acute coronary syndrome is recommended for patients with hemodynamic compromise, ongoing ischemia, or inadequate rate control. |
| 2a | C-LD | 3. For synchronized cardioversion of atrial fibrillation using biphasic energy, an initial energy of 120 to 200 J is reasonable, depending on the specific biphasic defibrillator being used. |
| 2b | C-LD | 4. For synchronized cardioversion of atrial flutter using biphasic energy, an initial energy of 50 to 100 J may be reasonable, depending on the specific biphasic defibrillator being used. |
Recommendation-Specific Supportive Text
- and 2. Uncontrolled tachycardia may impair ventricular filling, cardiac output, and coronary perfusion while increasing myocardial oxygen demand. While an expeditious trial of medications and/or fluids may be appropriate in some cases, unstable patients or patients with ongoing cardiac ischemia with atrial fibrillation or atrial flutter need to be cardioverted promptly.1–3 When making the decision for cardioversion, one should also consider whether the arrhythmia is the cause of the tachycardia. Potential exacerbation of rapid ventricular response by secondary causes (eg, sepsis) should be considered and may inform initial attempts at hemodynamic stabilization with pharmacotherapy. There are few data addressing these strategies in hemodynamically unstable patients. However, studies demonstrating hemodynamic benefits of successful cardioversion have been published.4,5 In addition, risks of hypotension and hypoperfusion with use of negative inotropes have been demonstrated even in normotensive patients.6–8 Hemodynamically unstable patients and those with ongoing cardiac ischemia are likely to benefit from the improved hemodynamic status associated with restoration of sinus rhythm and avoidance of hypotension caused by the alternative pharmacological therapies. Depending on the clinical scenario, patients cardioverted from atrial fibrillation or atrial flutter of 48 hours’ duration or longer are candidates for anticoagulation. Details about anticoagulation selection can be found elsewhere.2
- and 4. The electric energy required to successfully cardiovert a patient from atrial fibrillation or atrial flutter to sinus rhythm varies and is generally less in patients with new-onset arrhythmia, thin body habitus, and when biphasic waveform shocks are delivered.9–15 Obese patients may require greater energy.16 If initial cardioversion is unsuccessful, energy is increased in subsequent attempts. Less energy is generally required for atrial flutter than for atrial fibrillation.11 Higher energies of 200 J or more are associated with improved first shock success and decreased total energy delivery. In addition, a retrospective analysis found that lower energy shocks were associated with higher risk of cardioversion-induced VF.17 Previous guidelines included a comparison of monophasic and biphasic waveforms. This recommendation now focuses primarily on biphasic waveforms. Recommended energy levels vary with different devices, reducing the validity of generalized recommendations. This topic requires further study with a comprehensive systematic review to better understand the optimal electric doses with current devices. The writing group assessment of the LOE as C-LD is consistent with the limited evidence using modern devices and energy waveforms.
These recommendations are supported by the “2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/AHA Task Force on Practice Guidelines and the Heart Rhythm Society”18 as well as the focused update of those guidelines published in 2019.2
Open table in a new window.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. IV administration of a β-adrenergic blocker or nondihydropyridine calcium channel antagonist is recommended to slow the ventricular heart rate in the acute setting in patients with atrial fibrillation or atrial flutter with rapid ventricular response without preexcitation. |
| 2a | B-NR | 2. IV amiodarone can be useful for rate control in critically ill patients with atrial fibrillation with rapid ventricular response without preexcitation. |
| 3: Harm | C-LD | 3. In patients with atrial fibrillation and atrial flutter in the setting of preexcitation, digoxin, nondihydropyridine calcium channel antagonists, β-adrenergic blockers, and IV amiodarone should not be administered because they may increase the ventricular response and result in VF. |
| 3: Harm | C-EO | 4. Nondihydropyridine calcium channel antagonists and IV β-adrenergic blockers should not be used in patients with left ventricular systolic dysfunction and decompensated heart failure because these may lead to further hemodynamic compromise. |
Recommendation-Specific Supportive Text
- and 2. Clinical trial evidence shows that nondihydropyridine calcium channel antagonists (eg, diltiazem, verapamil), β-adrenergic blockers (eg, esmolol, propranolol), amiodarone, and digoxin are all effective for rate control in patients with atrial fibrillation/ flutter.6–8,19–23 Calcium channel blockers may be more effective than amiodarone, and cause more hypotension.6 Digoxin is rarely used in the acute setting because of slow onset of effect.1,2
- Based on limited case reports and small case series, there is concern that patients with concomitant preexcitation and atrial fibrillation or atrial flutter may develop VF in response to accelerated ventricular response after the administration of AV nodal blocking agents such as digoxin, nondihydropyridine calcium channel antagonists, β-adrenergic blockers, or IV amiodarone.24–27 In this setting, cardioversion is recommended as the most appropriate management.
- Because of their negative inotropic effect, nondihydropyridine calcium channel antagonists (eg, diltiazem, verapamil) may further decompensate patients with left ventricular systolic dysfunction and symptomatic heart failure. They may be used in patients with heart failure with preserved ejection fraction. β-Adrenergic blockers may be used in compensated patients with cardiomyopathy; however, they should be used with caution or avoided altogether in patients with decompensated heart failure. This recommendation is based on expert consensus and pathophysiologic rationale.2,18,28 β-Adrenergic blockers may be used in patients with chronic obstructive pulmonary disease because multiple studies have shown no negative effects.29
These recommendations are supported by 2014 AHA, American College of Cardiology, and Heart Rhythm Society Guideline for the Management of Patients With Atrial Fibrillation18 as well as the focused update of those guidelines published in 2019.2
Introduction
Bradycardia is generally defined as a heart rate less than 60/min. Bradycardia can be a normal finding, especially for athletes or during sleep. When bradycardia occurs secondary to a pathological cause, it can lead to decreased cardiac output with resultant hypotension and tissue hypoperfusion. The clinical manifestations of bradycardia can range from an absence of symptoms to symptomatic bradycardia (bradycardia associated with acutely altered mental status, ischemic chest discomfort, acute heart failure, hypotension, or other signs of shock that persist despite adequate airway and breathing). The cause of the bradycardia may dictate the severity of the presentation. For example, patients with severe hypoxia and impending respiratory failure may suddenly develop a profound bradycardia that leads to cardiac arrest if not addressed immediately. In contrast, a patient who develops third-degree heart block but is otherwise well compensated might experience relatively low blood pressure but otherwise be stable. Therefore, the management of bradycardia will depend on both the underlying cause and severity of the clinical presentation. In 2018, the AHA, American College of Cardiology, and Heart Rhythm Society published an extensive guideline on the evaluation and management of stable and unstable bradycardia.2 This guideline focuses exclusively on symptomatic bradycardia in the ACLS setting and maintains consistency with the 2018 guideline.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-EO | 1. In patients presenting with acute symptomatic bradycardia, evaluation and treatment of reversible causes is recommended. |
| 2a | B-NR | 2. In patients with acute bradycardia associated with hemodynamic compromise, administration of atropine is reasonable to increase heart rate. |
| 2b | C-LD | 3. If bradycardia is unresponsive to atropine, IV adrenergic agonists with rate-accelerating effects (eg, epinephrine) or transcutaneous pacing may be effective while the patient is prepared for emergent transvenous temporary pacing if required. |
| 2b | C-EO | 4. Immediate pacing might be considered in unstable patients with high-degree AV block when IV/IO access is not available. |
Recommendation-Specific Supportive Text
- Symptomatic bradycardia may be caused by a number of potentially reversible or treatable causes, including structural heart disease, increased vagal tone, hypoxemia, myocardial ischemia, or medications.2 Bradycardia may be difficult to resolve until the underlying cause is treated, making evaluation of underlying cause imperative, simultaneous with emergent treatments for stabilization.
- Atropine has been shown to be effective for the treatment of symptomatic bradycardia in both observational studies and in 1 limited RCT.3–7
- If atropine is ineffective, either alternative agents to increase heart rate and blood pressure or transcutaneous pacing are reasonable next steps. For medical management of a periarrest patient, epinephrine has gained popularity, including IV infusion and utilization of “push-dose” administration for acute bradycardia and hypotension. Studies on push-dose epinephrine for bradycardia specifically are lacking, although limited data support its use for hypotension.8 Use of push-dose vasopressor requires careful attention to correct dosing. Medication errors leading to adverse effects have been reported.9 Dopamine infusion can also increase heart rate.10 There are limited studies comparing medications to transcutaneous pacing for the treatment of bradycardia. A randomized feasibility study in patients failing atropine compared dopamine to transcutaneous pacing and found no difference in survival to discharge.10 Whether to trial transcutaneous pacing, epinephrine, dopamine, or other vasoactive agent will likely therefore depend on clinician experience and resources available.
- For severe symptomatic bradycardia causing shock, if no IV or IO access is available, immediate transcutaneous pacing while access is being pursued may be undertaken. A 2006 systematic review involving 7 studies of transcutaneous pacing for symptomatic bradycardia and bradyasystolic cardiac arrest in the prehospital setting did not find a benefit from pacing compared with standard ACLS, although a subgroup analysis from 1 trial suggested a possible benefit in patients with symptomatic bradycardia.11
These recommendations are supported by the “2018 ACC/AHA/HRS Guideline on the Evaluation and Management of Patients With Bradycardia and Cardiac Conduction Delay: A Report of the American College of Cardiology/AHA Task Force on Clinical Practice Guidelines and the Heart Rhythm Society.”2
| COR | LOE | Recommendation |
|---|---|---|
| 2a | C-LD | 1. In patients with persistent hemodynamically unstable bradycardia refractory to medical therapy, temporary transvenous pacing is reasonable to increase heart rate and improve symptoms. |
Recommendation-Specific Supportive Text
- When bradycardia is refractory to medical management and results in severe symptoms, the reasonable next step is placement of a temporary pacing catheter for transvenous pacing. Limited evidence for this intervention consists largely of observational studies, many of which have focused on indications and the relatively high complication rate (including bloodstream infections and pneumothorax, among others).12–14 However, when the heart rate does not improve with medications and shock persists, transvenous pacing can improve the heart rate and symptoms until more definitive treatment (correction of underlying cause or permanent pacemaker placement) can be implemented.
These recommendations are supported by the 2018 American College of Cardiology, AHA, and Heart Rhythm Society guideline on the evaluation and management of patients with bradycardia and cardiac conduction delay.2
Introduction
Post–cardiac arrest care is a critical component of the Chain of Survival. What defines optimal hospital care for patients with ROSC after cardiac arrest is not completely known, but there is increasing interest in identifying and optimizing practices that are likely to improve outcomes. The systemic impact of the ischemia-reperfusion injury caused by cardiac arrest and subsequent resuscitation requires post–cardiac arrest care to simultaneously support the multiple organ systems that are affected. After initial stabilization, care of critically ill postarrest patients hinges on hemodynamic support, mechanical ventilation, temperature management, diagnosis and treatment of underlying causes, diagnosis and treatment of seizures, vigilance for and treatment of infection, and management of the critically ill state of the patient. Many cardiac arrest patients who survive the initial event will eventually die because of withdrawal of life-sustaining treatment in the setting of neurological injury. This cause of death is especially prominent in those with OHCA but is also frequent after IHCA.1,2 Thus, much of postarrest care focuses on mitigating injury to the brain. Possible contributors to this goal include optimization of cerebral perfusion pressure, management of oxygen and carbon dioxide levels, control of core body temperature, and detection and treatment of seizures (Figure 9). Cardiac arrest results in heterogeneous injury; thus, death can also result from multiorgan dysfunction or shock. In light of the complexity of postarrest patients, a multidisciplinary team with expertise in cardiac arrest care is preferred, and the development of multidisciplinary protocols is critical to optimize survival and neurological outcome.
Key topics in postresuscitation care that are not covered in this section, but are discussed later, are targeted temperature management (TTM) (Targeted Temperature Management), percutaneous coronary intervention (PCI) in cardiac arrest (PCI After Cardiac Arrest), neuroprognostication (Neuroprognostication), and recovery (Recovery).
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. A comprehensive, structured, multidisciplinary system of care should be implemented in a consistent manner for the treatment of post–cardiac arrest patients. |
| 1 | B-NR | 2. A 12-lead ECG should be obtained as soon as feasible after ROSC to determine whether acute ST-segment elevation is present. |
| 2a | C-EO | 3. To avoid hypoxia in adults with ROSC in the immediate postarrest period, it is reasonable to use the highest available oxygen concentration until the arterial oxyhemoglobin saturation or the partial pressure of arterial oxygen can be measured reliably. |
Recommendation-Specific Supportive Text
- Observational studies evaluating the utility of cardiac receiving centers suggest that a strong system of care may represent a logical clinical link between successful resuscitation and ultimate survival.3 Although data are limited, taken together with experience from regionalized approaches to other emergencies such as trauma, stroke, and ST-segment elevation acute myocardial infarction, consistent implementation of a system of care to manage cardiac arrest patients may improve outcomes.
- Patients with 12-lead identification of ST-segment elevation myocardial infarction (STEMI) should have coronary angiography for possible PCI, highlighting the importance of obtaining an ECG for diagnostic purposes.4 However, multiple studies have reported that absence of ST-segment elevations does not rule out an intervenable coronary lesion.5–7
- Several RCTs have compared a titrated approach to oxygen administration with an approach of administering 100% oxygen in the first 1 to 2 hours after ROSC.8–10 All of these were conducted in the prehospital setting. However, these trials only titrated oxygen once an oxygen saturation could be measured with a pulse oximeter. No studies have investigated titration of oxygen in patients for whom oxygen saturation (by pulse oximeter) or partial pressure of oxygen in the blood (by arterial blood gas) cannot be measured. The recommendation to administer 100% oxygen until measurement of this vital sign is possible is therefore based on physiology and the expert opinion that hypoxia could worsen end-organ damage and should be avoided.
Recommendation 1 is supported by the 2019 focused update on ACLS guidelines.3 Recommendation 2 last received formal evidence review in 2015.4 Recommendation 3 is supported by the 2020 CoSTR for ALS.11
| COR | LOE | Recommendation |
|---|---|---|
| 2a | B-NR | 1. It is preferable to avoid hypotension by maintaining a systolic blood pressure of at least 90 mm Hg and a mean arterial pressure of at least 65 mm Hg in the postresuscitation period. |
Recommendation-Specific Supportive Text
- Hypotension may worsen brain and other organ injury after cardiac arrest by decreasing oxygen delivery to tissues. The optimal MAP target after ROSC, however, is not clear. This topic was previously reviewed by ILCOR in 2015,12 and a detailed evidence update was conducted by the Australia and New Zealand Council of Resuscitation on behalf of ILCOR for 2020.11 Several observational studies have found that postresuscitation hypotension is associated with worse survival and neurological outcome.13–19 One study found no association between higher MAP during TTM treatment and outcome, although shock at admission was associated with poor outcome.20 Definitions of hypotension vary between studies, with systolic blood pressure of 90 mm Hg and MAP of 65 mm Hg being common cutoffs used. Two RCTs conducted since 2015 compared a lower blood pressure target (standard care or MAP greater than 65 mm Hg in one study and MAP 65–75 mm Hg in the other) with a higher target (MAP 85–100 in one study and MAP 80–100 mm Hg in the other).21,22 Both studies failed to detect any difference in survival or survival with favorable neurological outcome, although neither study was appropriately powered for these outcomes. One trial did find improvement in cerebral oxygenation with higher MAP,21 which is a proposed mechanism for the benefit effect of higher MAP in hypoxic ischemic encephalopathy. A recent observational study comparing outcomes in patients with MAP 70 to 90 mm Hg to those with MAP greater than 90 mm Hg also found that higher MAP was associated with better neurological outcome.23 Although some of these data suggest targeting a MAP of 80 mm Hg or higher in those at risk for neurological injury after cardiac arrest might be beneficial, this remains unproven.
These recommendations are supported by the 2015 Guidelines Update24 and a 2020 evidence update.11
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend avoiding hypoxemia in all patients who remain comatose after ROSC. |
| 2b | B-R | 2. Once reliable measurement of peripheral blood oxygen saturation is available, avoiding hyperoxemia by titrating the fraction of inspired oxygen to target an oxygen saturation of 92% to 98% may be reasonable in patients who remain comatose after ROSC. |
| 2b | B-R | 3. Maintaining the arterial partial pressure of carbon dioxide (Paco2) within a normal physiological range (generally 35–45 mm Hg) may be reasonable in patients who remain comatose after ROSC. |
Recommendation-Specific Supportive Text
- In a 2020 ILCOR systematic review,11 1 observational study reported that hypoxemia after return of circulation was associated with worse outcome.25 This was not seen in other studies,26–28 and all studies were at high risk of bias. This recommendation is therefore based primarily on the physiological rationale that hypoxia increases the risk of end-organ damage, and the fact that hypoxemia is the best available surrogate for hypoxia.
- There are some physiological basis and preclinical data for hyperoxemia leading to increased inflammation and exacerbating brain injury in post-arrest patients.29 A 2020 ILCOR systematic review11 identified 5 RCTs comparing a titrated or lower oxygen administration strategy with usual care or a higher oxygen administration strategy in post-arrest patients: 3 in the prehospital setting and 2 in the ICU setting.8–10,30,31 Overall, these trials found no difference in clinical outcomes, but all were underpowered for these outcomes. A recent large RCT compared usual care with aggressive avoidance of hyperoxemia in mechanically ventilated critically ill patients and found no difference between groups in the overall cohort but increased survival in the intervention arm in the subgroup of 164 post-arrest patients.32 Observational data are inconsistent and very limited by confounding.11 Three RCTs on this topic are ongoing (NCT03138005, NCT03653325, NCT03141099). The suggested range of 92% to 98% is intended as a practical approximation of the normal range.
- Two RCTs compared a strategy of targeting high-normal Paco2 (44–46 mmHg) with one targeting low-normal Paco2 (33–35 mmHg)31 and a strategy targeting moderate hypercapnia (Paco2 50– 55 mmHg) compared with normocapnia (Paco2 35–45 mmHg).33 Neither trial found a difference in any clinical outcomes. Results across 6 observational studies were inconsistent, and all studies were limited by significant risk of bias.25,34–38 There is a large ongoing RCT addressing this question (NCT03114033).
These recommendations are supported by the 2020 CoSTR for ALS.11
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. We recommend treatment of clinically apparent seizures in adult post–cardiac arrest survivors. |
| 1 | C-LD | 2. We recommend promptly performing and interpreting an electroencephalogram (EEG) for the diagnosis of seizures in all comatose patients after ROSC. |
| 2b | C-LD | 3. The treatment of nonconvulsive seizures (diagnosed by EEG only) may be considered. |
| 2b | C-LD | 4. The same anticonvulsant regimens used for the treatment of seizures caused by other etiologies may be considered for seizures detected after cardiac arrest. |
| 3: No Benefit | B-R | 5. Seizure prophylaxis in adult post–cardiac arrest survivors is not recommended. |
Recommendation-Specific Supportive Text
- A 2020 ILCOR systematic review11 identified no controlled studies comparing treatment of seizures with no treatment of seizures in this population. In spite of the lack of evidence, untreated clinically apparent seizure activity is thought to be potentially harmful to the brain, and treatment of seizures is recommended in other settings39 and likely also warranted after cardiac arrest.
- The writing group acknowledged that there is no direct evidence that EEG to detect nonconvulsive seizures improves outcomes. This recommendation is based on the fact that nonconvulsive seizures are common in postarrest patients and that the presence of seizures may be important prognostically, although whether treatment of nonconvulsive seizures affects outcome in this setting remains uncertain. An ILCOR systematic review done for 2020 did not specifically address the timing and method of obtaining EEGs in postarrest patients who remain unresponsive. Data on the relative benefit of continuous versus intermittent EEG are limited. One study found no difference in survival with good neurological outcome at 3 months in patients monitored with routine (one to two 20-minute EEGs over 24 hours) versus continuous (for 18–24 hours) EEG.40
- Nonconvulsive seizures are common after cardiac arrest. Whether treatment of seizure activity on EEG that is not associated with clinically evident seizures affects outcome is currently unknown. A randomized trial investigating this question is ongoing (NCT02056236).
- The 2020 CoSTR recommends that seizures be treated when diagnosed in postarrest patients.11 No specific agent was recommended. However, the CoSTR described 2 retrospective studies suggesting valproate, levetiracetam, and fosphenytoin may all be effective, with fosphenytoin found to be associated with more hypotension in 1 study.41,42 Common sedatives such as propofol and midazolam have also been found to be effective in suppressing seizure activity after cardiac arrest.43–45
- A 2020 ILCOR systematic review11 identified 2 RCTs comparing seizure prophylaxis with no seizure prophylaxis in comatose postarrest patients.46,47 Neither study found any difference in occurrence of seizures or survival with favorable neurological outcome between groups.
These recommendations are supported by the 2020 CoSTR for ALS.11
Recommendation-Specific Supportive Text
- A 2020 ILCOR systematic review11 identified no controlled studies comparing treatment of seizures with no treatment of seizures in this population. In spite of the lack of evidence, untreated clinically apparent seizure activity is thought to be potentially harmful to the brain, and treatment of seizures is recommended in other settings39 and likely also warranted after cardiac arrest.
- The writing group acknowledged that there is no direct evidence that EEG to detect nonconvulsive seizures improves outcomes. This recommendation is based on the fact that nonconvulsive seizures are common in postarrest patients and that the presence of seizures may be important prognostically, although whether treatment of nonconvulsive seizures affects outcome in this setting remains uncertain. An ILCOR systematic review done for 2020 did not specifically address the timing and method of obtaining EEGs in postarrest patients who remain unresponsive. Data on the relative benefit of continuous versus intermittent EEG are limited. One study found no difference in survival with good neurological outcome at 3 months in patients monitored with routine (one to two 20-minute EEGs over 24 hours) versus continuous (for 18–24 hours) EEG.40
- Nonconvulsive seizures are common after cardiac arrest. Whether treatment of seizure activity on EEG that is not associated with clinically evident seizures affects outcome is currently unknown. A randomized trial investigating this question is ongoing (NCT02056236).
- The 2020 CoSTR recommends that seizures be treated when diagnosed in postarrest patients.11 No specific agent was recommended. However, the CoSTR described 2 retrospective studies suggesting valproate, levetiracetam, and fosphenytoin may all be effective, with fosphenytoin found to be associated with more hypotension in 1 study.41,42 Common sedatives such as propofol and midazolam have also been found to be effective in suppressing seizure activity after cardiac arrest.43–45
- A 2020 ILCOR systematic review11 identified 2 RCTs comparing seizure prophylaxis with no seizure prophylaxis in comatose postarrest patients.46,47 Neither study found any difference in occurrence of seizures or survival with favorable neurological outcome between groups.
These recommendations are supported by the 2020 CoSTR for ALS.11
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-R | 1. The benefit of any specific target range of glucose management is uncertain in adults with ROSC after cardiac arrest. |
| 2b | B-R | 2. The routine use of prophylactic antibiotics in postarrest patients is of uncertain benefit. |
| 2b | B-R | 3. The effectiveness of agents to mitigate neurological injury in patients who remain comatose after ROSC is uncertain. |
| 2b | B-R | 4. The routine use of steroids for patients with shock after ROSC is of uncertain value. |
Recommendation-Specific Supportive Text
- One small RCT from 2007,48 found no difference in survival between strict and moderate glucose control. In the absence of other evidence specific to cardiac arrest, it seems reasonable to manage blood glucose levels in postarrest patients with the same approach used for the general critically ill population, namely using insulin therapy when needed to maintain a blood glucose of 150 to 180 mg/dL.49
- A 2020 ILCOR systematic review found 2 RCTs and a small number of observational studies evaluating the effect of prophylactic antibiotics on outcomes in postarrest patients.11,50 The RCTs found no difference in survival or neurological outcome.51,52 One RCT51 did find lower incidence of early pneumonia in those who received prophylactic antibiotics, but this did not translate to a difference in other outcomes. When data from the 2 RCTs were pooled, there was no overall difference in infections.51,52
- The topic of neuroprotective agents was last reviewed in detail in 2010. Multiple agents, including magnesium, coenzyme Q10 (ubiquinol), exanatide, xenon gas, methylphenidate, and amantadine, have been considered as possible agents to either mitigate neurological injury or facilitate patient awakening. This work has been largely observational,53–57 although randomized trials have been conducted on coenzyme Q10, xenon gas, and exanatide.58–60 A small trial on the effect of coenzyme Q10 reported better survival in those receiving coenzyme Q10, but there was no significant difference in favorable neurological outcome and these findings have yet to be validated.58 One additional coenzyme Q10 trial was recently completed but results are not yet available (NCT02934555). None of the other studies identified have been able to show a difference in any clinical outcomes with use of any of the agents studied.
- Since this topic was last updated in detail in 2015, at least 2 randomized trials have been completed on the effect of steroids on shock and other outcomes after ROSC, only 1 of which has been published to date.61 In this study, shock reversal and other outcomes did not differ between groups. A large retrospective observational study did find that steroid use after cardiac arrest was associated with survival.62 Steroid use for septic shock has been evaluated extensively, with a recent trial of over 1200 patients finding improved survival in those treated with steroids.63 A trial enrolling 3800 patients did not find a mortality benefit, although time to discharge from ICU and time to shock reversal were both shorter in the steroid group.64 Taken together, there is no definitive evidence of benefit from steroids after ROSC. However, the data in sepsis suggest that some patients with severe shock may benefit from steroids and that the co-occurrence of sepsis and cardiac arrest is important to consider.
Recommendation 1 last received formal evidence review in 2010 and is supported by the “Guidelines for the Use of an Insulin Infusion for the Management of Hyperglycemia in Critically Ill Patients” from the Society for Critical Care Medicine.49 Recommendation 2 is supported by the 2020 CoSTR for ALS.11 Recommendations 3 and 4 last received formal evidence review in 2015.24
Introduction
TTM between 32°C and 36°C for at least 24 hours is currently recommended for all cardiac rhythms in both OHCA and IHCA. Multiple randomized trials have been performed in various domains of TTM and were summarized in a systematic review published in 2015.1 Subsequent to the 2015 recommendations, additional randomized trials have evaluated TTM for nonshockable rhythms as well as TTM duration. Many of these were reviewed in an evidence update provided in the 2020 COSTR for ALS.2 Many uncertainties within the topic of TTM remain, including whether temperature should vary on the basis of patient characteristics, how long TTM should be maintained, and how quickly it should be started. An updated systematic review on several aspects of this important topic is needed once currently ongoing clinical trials have been completed.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-R | 1. We recommend TTM for adults who do not follow commands after ROSC from OHCA with any initial rhythm. |
| 1 | B-R | 2. We recommend TTM for adults who do not follow commands after ROSC from IHCA with initial nonshockable rhythm. |
| 1 | B-NR | 3. We recommend TTM for adults who do not follow commands after ROSC from IHCA with initial shockable rhythm. |
Recommendation-Specific Supportive Text
- Two RCTs of patients with OHCA with an initially shockable rhythm published in 2002 reported benefit from mild hypothermia when compared with no temperature management.1,3,4 A more recent trial comparing a target temperature of 33°C to 37°C in patients (IHCA and OHCA) with initial nonshockable rhythm also found better outcomes in those treated with a temperature of 33°C.5 A large trial is currently underway testing TTM compared with normothermia (NCT03114033).
- An RCT published in 2019 compared TTM at 33°C to 37°C for patients who were not following commands after ROSC from cardiac arrest with initial nonshockable rhythm. Survival with a favorable neurological outcome (Cerebral Performance Category 1–2) was higher in the group treated with 33°C.5 This trial included both OHCA and IHCA and is the first randomized trial on TTM after cardiac arrest to include IHCA patients. In a subgroup analysis, the benefit of TTM did not appear to differ significantly by IHCA/OHCA subgroups.
- No RCTs of TTM have included IHCA patients with an initial shockable rhythm, and this recommendation is therefore based largely on extrapolation from OHCA studies and the study of patients with initially nonshockable rhythms that included IHCA patients. Observational studies on TTM for IHCA with any initial rhythm have reported mixed results. Two studies that included patients enrolled in the AHA Get With The Guidelines Resuscitation registry reported either no benefit or worse outcome from TTM.6,7 Both were limited by very low overall usage of TTM in the registry and lack of data on presence of coma, making it difficult to determine if TTM was indicated for a given IHCA patient.
This topic last received formal evidence review in 2015,8 with an evidence update conducted for the 2020 CoSTR for ALS.2
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-R | 1. We recommend selecting and maintaining a constant temperature between 32°C and 36°C during TTM. |
| 2a | B-NR | 2. It is reasonable that TTM be maintained for at least 24 h after achieving target temperature. |
| 2b | C-LD | 3. It may be reasonable to actively prevent fever in comatose patients after TTM. |
| 3: No Benefit | A | 4. We do not recommend the routine use of rapid infusion of cold IV fluids for prehospital cooling of patients after ROSC |
Recommendation-Specific Supportive Text
- In 2013, a trial of over 900 patients compared TTM at 33°C to 36°C for patients with OHCA and any initial rhythm, excluding unwitnessed asystole, and found that 33°C was not superior to 36°C.9 A more recent trial compared 33°C to 37°C for patients with ROSC after initial nonshockable rhythm and found improved survival with favorable neurological outcome in the group treated with 33°C.5 There have been reports of decreasing utilization of TTM in recent years, with one hypothesis being that some clinicians interpret the inclusion of 36°C as a target temperature as being equivalent to normothermia, or no strict temperature control.10 An updated systematic review is needed on the question of which target temperature is most beneficial. Based on the available evidence, however, TTM at a temp between 32°C and 36°C remains a Class 1 recommendation.
- One RCT including 355 patients found no difference in outcome between TTM for 24 and 48 hours.11 This study may have been underpowered to detect differences in clinical outcomes. The initial 2002 trials cooled patients for 123 and 24 hours4 while the 2013 trial used 28 hours.9 A larger, adaptive clinical trial is currently underway investigating multiple different durations of hypothermia ranging from 6 to 72 hours, using a target temperature of 33°C for all patients enrolled (NCT04217551). There is no clear best approach to rewarming after TTM, although a protocol of 0.5°C per hour was followed in the 2013 trial.9 The optimal rate of rewarming, and specifically whether slower rates are beneficial, is a knowledge gap, and at least 1 trial is ongoing (NCT02555254).
- Fever after ROSC is associated with poor neurological outcome in patients not treated with TTM, although this finding is reported less consistently in patients treated with TTM.12–20 It has not been established whether treatment of fever s associated with an improvement in outcome, but treatment or prevention of fever appears to be a reasonable approach.
- A 2015 systematic review found that prehospital cooling with the specific method of the rapid infusion of cold IV fluids was associated with more pulmonary edema and a higher risk of rearrest.1 Since this review, a number of RCTs on prehospital cooling have been conducted. One trial compared the prehospital induction of hypothermia with any method (including ice packs and cold IV fluids) with no prehospital cooling, and found higher receipt of in-hospital TTM in those who had prehospital initiation. That trial found no increased adverse events in those treated with prehospital cooling.21 Other methods of prehospital cooling, such as esophageal or nasal devices, have also been investigated; whether these affect outcomes is a knowledge gap.
This topic last received formal evidence review in 2015,8 with an evidence update conducted for the 2020 CoSTR for ALS.2
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. Coronary angiography should be performed emergently for all cardiac arrest patients with suspected cardiac cause of arrest and ST-segment elevation on ECG. |
| 2a | B-NR | 2. Emergent coronary angiography is reasonable for select (eg, electrically or hemodynamically unstable) adult patients who are comatose after OHCA of suspected cardiac origin but without ST-segment elevation on ECG. |
| 2a | C-LD | 3. Independent of a patient’s mental status, coronary angiography is reasonable in all post–cardiac arrest patients for whom coronary angiography is otherwise indicated. |
Synopsis
Coronary artery disease (CAD) is prevalent in the setting of cardiac arrest.1–4 Patients with cardiac arrest due to shockable rhythms have demonstrated particularly high rates of severe CAD: up to 96% of patients with STEMI on their postresuscitation ECG,2,5 up to 42% for patients without ST-segment elevation,2,5–7 and 85% of refractory out-of-hospital VF/VT arrest patients have severe CAD.8 The role of CAD in cardiac arrest with nonshockable rhythms is unknown.
When significant CAD is observed during post-ROSC coronary angiography, revascularization can be achieved safely in most cases.5,7,9 Further, successful PCI is associated with improved survival in multiple observational studies.2,6,7,10,11 Additional benefits of evaluation in the cardiac catheterization laboratory include discovery of anomalous coronary anatomy, the opportunity to assess left ventricular function and hemodynamic status, and the potential for insertion of temporary mechanical circulatory support devices.
The 2015 Guidelines Update recommended emergent coronary angiography for patients with ST-segment elevation on the post-ROSC ECG. Emergent coronary angiography and PCI have also been also associated with improved neurological outcomes in patients without STEMI on their post-ROSC resuscitation ECG.4,12 However, a large randomized trial found no improvement in survival in patients resuscitated from OHCA with an initial shockable rhythm in whom no ST-segment elevations or signs of shock were present.13 Multiple RCTs are underway. It remains to be tested whether patients with signs of shock benefit from emergent coronary angiography and PCI.
Recommendation-Specific Supportive Text
- Several observational studies have demonstrated improved neurologically favorable survival when early coronary angiography is performed followed by PCI in patients with cardiac arrest who have a STEMI.5,14–17 This led to a Class 1 recommendation in the 2015 Guidelines Update that has not been contradicted by any other recent studies. This recommendation is consistent with global recommendations for all patients with STEMI.
- Multiple observational studies have shown an association between emergent coronary angiography and PCI and improved neurological outcomes in patients without ST-segment elevation.5,7,14,15,18 A meta-analysis also supported the use of early coronary angiography in patients without ST-segment elevation.19 However, a large randomized trial found no improvement in survival in patients resuscitated from OHCA with an initial shockable rhythm in whom no ST-segment elevation or signs of shock were present.20 In addition, while coronary artery disease was found in 65% of patients who underwent coronary angiography, only 5% of patients had acute thrombotic coronary occlusions. Multiple RCTs are underway, but the role of emergent coronary angiography and PCI in patients without ST-elevation but with signs of shock remains to be tested. The use of emergent coronary angiography in patients with hemodynamic or electric instability is consistent with guidelines for non-STEMI patients.21–23 The optimal treatment of hemodynamically and electrically stable patients without ST-segment elevation remains unclear. This area was last reviewed systematically in 2015 and requires additional systematic review after the completion of currently active trials (NCT03119571, NCT02309151, NCT02387398, NCT02641626, NCT02750462, NCT02876458).
- Evidence suggests that patients who are comatose after ROSC benefit from invasive angiography, when indicated, as do patients who are awake.4,14,18 Therefore, invasive coronary angiography is reasonable independent of neurological status.
This topic last received formal evidence review in 2015.24
General Considerations for Neuroprognostication Introduction
Hypoxic-ischemic brain injury is the leading cause of morbidity and mortality in survivors of OHCA and accounts for a smaller but significant portion of poor outcomes after resuscitation from IHCA.1,2 Most deaths attributable to postarrest brain injury are due to active withdrawal of life-sustaining treatment based on a predicted poor neurological outcome. Accurate neurological prognostication is important to avoid inappropriate withdrawal of life-sustaining treatment in patients who may otherwise achieve meaningful neurological recovery and also to avoid ineffective treatment when poor outcome is inevitable (Figure 10).3
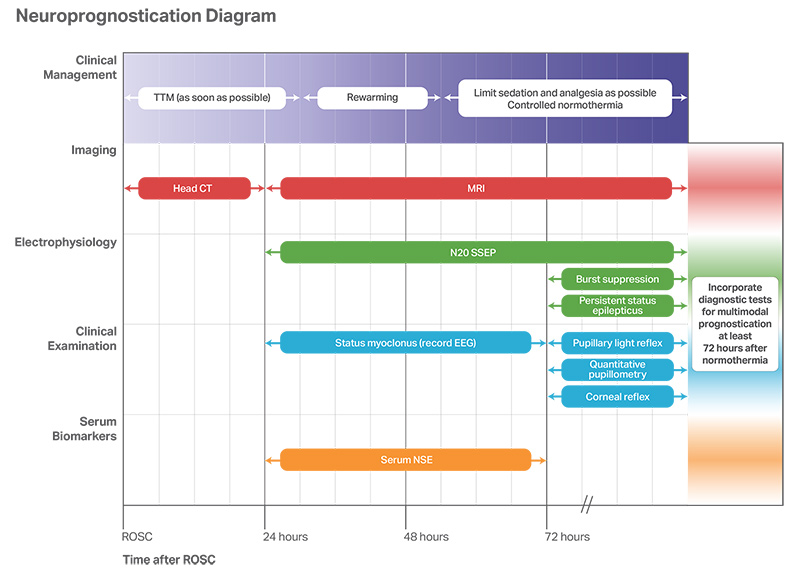
Neurologic prognostication incorporates multiple diagnostic tests which are synthesized into a comprehensive multimodal assessment at least 72 hours after return to normothermia and with sedation and analgesia limited as possible. Awareness and incorporation of the potential sources of error in the individual diagnostic tests is important. The suggested timing of the multimodal diagnostics is shown here. CT indicates computed tomography; EEG, electroencephalogram; MRI, magnetic resonance imaging; NSE, neuron-specific enolase; ROSC, return of spontaneous circulation; SSEP, somatosensory evoked potential; and TTM, targeted temperature management.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. In patients who remain comatose after cardiac arrest, we recommend that neuroprognostication involve a multimodal approach and not be based on any single finding. |
| 1 | B-NR | 2. In patients who remain comatose after cardiac arrest, we recommend that neuroprognostication be delayed until adequate time has passed to ensure avoidance of confounding by medication effect or a transiently poor examination in the early postinjury period. |
| 1 | C-EO | 3. We recommend that teams caring for comatose cardiac arrest survivors have regular and transparent multidisciplinary discussions with surrogates about the anticipated time course for and uncertainties around neuroprognostication. |
| 2a | B-NR | 4. In patients who remain comatose after cardiac arrest, it is reasonable to perform multimodal neuroprognostication at a minimum of 72 hours after normothermia, though individual prognostic tests may be obtained earlier than this. |
Synopsis
Neuroprognostication relies on interpreting the results of diagnostic tests and correlating those results with outcome. Given that a false-positive test for poor neurological outcome could lead to inappropriate withdrawal of life support from a patient who otherwise would have recovered, the most important test characteristic is specificity. Many of the tests considered are subject to error because of the effects of medications, organ dysfunction, and temperature. Furthermore, many research studies have methodological limitations including small sample sizes, single-center design, lack of blinding, the potential for self-fulfilling prophecies, and the use of outcome at hospital discharge rather than a time point associated with maximal recovery (typically 3–6 months after arrest).3
Because any single method of neuroprognostication has an intrinsic error rate and may be subject to confounding, multiple modalities should be used to improve decision-making accuracy.
Recommendation-Specific Supportive Text
- The overall certainty in the evidence of neurological prognostication studies is low because of biases that limit the internal validity of the studies as well as issues of generalizability that limit their external validity. Thus, the confidence in the prognostication of the diagnostic tests studied is also low. Neuroprognostication that uses multimodal testing is felt to be better at predicting outcomes than is relying on the results of a single test to predict poor prognosis.3,4
- Sedatives and neuromuscular blockers may be metabolized more slowly in post–cardiac arrest patients, and injured brains may be more sensitive to the depressant effects of various medications. Residual sedation or paralysis can confound the accuracy of clinical examinations.5
- Prognostication of neurological recovery is complex and limited by uncertainty in most cases. Discordance in goals of care between clinicians and families/surrogates has been reported in more than 25% of critically ill patients.6 Lack of adequate communication is one important factor, and regular multidisciplinary conversations may help mitigate this.
- Operationally, the timing for prognostication is typically at least 5 days after ROSC for patients treated with TTM (which is about 72 hours after normothermia) and should be conducted under conditions that minimize the confounding effects of sedating medications. Individual test modalities may be obtained earlier and the results integrated into the multimodality assessment synthesized at least 72 hours after normothermia. In some instances, prognostication and withdrawal of life support may appropriately occur earlier because of nonneurologic disease, brain herniation, patient’s goals and wishes, or clearly nonsurvivable situations.
These recommendations are supported by the 2020 CoSTR for ALS,4 which supplements the last comprehensive review of this topic conducted in 2015.7
Use of the Clinical Examination in Neuroprognostication
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. When performed with other prognostic tests, it may be reasonable to consider bilaterally absent pupillary light reflex at 72 h or more after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | B-NR | 2. When performed with other prognostic tests, it may be reasonable to consider quantitative pupillometry at 72 h or more after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | B-NR | 3. When performed with other prognostic tests, it may be reasonable to consider bilaterally absent corneal reflexes at 72 h or more after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | B-NR | 4. When performed with other prognostic tests, it may be reasonable to consider status myoclonus that occurs within 72 h after cardiac arrest to support the prognosis of poor neurological outcome. |
| 2b | B-NR | 5. We suggest recording EEG in the presence of myoclonus to determine if there is an associated cerebral correlate. |
| 3: Harm | B-NR | 6. The presence of undifferentiated myoclonic movements after cardiac arrest should not be used to support a poor neurological prognosis. |
| 3: Harm | B-NR | 7. We recommend that the findings of a best motor response in the upper extremities being either absent or extensor movements not be used alone for predicting a poor neurological outcome in patients who remain comatose after cardiac arrest. |
Synopsis
Clinical examination findings correlate with poor outcome but are also subject to confounding by TTM and medications, and prior studies have methodological limitations. In addition to assessing level of consciousness and performing basic neurological examination, clinical examination elements may include the pupillary light reflex, pupillometry, corneal reflex, myoclonus, and status myoclonus when assessed within 1 week after cardiac arrest. The ILCOR systematic review included studies regardless of TTM status, and findings were correlated with neurological outcome at time points ranging from hospital discharge to 12 months after arrest.4 Quantitative pupillometry is the automated assessment of pupillary reactivity, measured by the percent reduction in pupillary size and the degree of reactivity reported as the neurological pupil index. Benefits of this method are a standard and reproducible assessment. Status myoclonus is commonly defined as spontaneous or sound-sensitive, repetitive, irregular brief jerks in both face and limb present most of the day within 24 hours after cardiac arrest.8 Status myoclonus differs from myoclonic status epilepticus; myoclonic status epilepticus is defined as status epilepticus with physical manifestation of persistent myoclonic movements and is considered a subtype of status epilepticus for these guidelines.
Recommendation-Specific Supportive Text
- In 17 studies,9–25 absent pupillary light reflex assessed from immediately after ROSC up to 7 days after arrest predicted poor neurological outcome with specificity ranging from 48% to 100%. The specificity varied significantly on the basis of timing, with the highest specificity seen at time points 72 hours or more after arrest.
- Three studies evaluated quantitative pupillary light reflex15,26,27 and 3 studies evaluated neurological pupil index15,28,29 at time points ranging from 24 to 72 hours after arrest. Absent pupillary light reflex as assessed by quantitative pupillometry (ie, quantitative pupillary light reflex=0%) is an objective finding and, in 1 study of 271 patients, had high specificity for poor outcome when assessed at 72 hours after arrest.15 Neurological pupil index is nonspecific and may be affected by medications; thus, an absolute neurological pupil index cutoff and a specific threshold that predicts poor prognosis is unknown.15,28,29
- Eleven observational studies9–11,14,16,17,19,21,22,30,31 evaluated absence of corneal reflexes at time points ranging from immediately after ROSC to 7 days after arrest. The specificity for poor outcome ranged from 25% to 100% and increased in the studies evaluating corneal reflexes at time points 72 hours or more after arrest (ranging from 89% to 100%). Like other examination findings, corneal reflexes are subject to confounding by medications, and few studies specifically evaluated the potential of residual medication effect.
- In 2 studies involving 347 patients,21,32 the presence of status myoclonus within 72 hours predicted poor neurological outcome from hospital discharge to 6 months, with specificity ranging from 97% to 100%.
- Obtaining EEG in status myoclonus is important to rule out underlying ictal activity. In addition, status myoclonus may have an EEG correlate that is not clearly ictal but may have prognostic meaning, and additional research is needed to delineate these patterns. Some EEG-correlated patterns of status myoclonus may have poor prognosis, but there may also be more benign subtypes of status myoclonus with EEG correlates.33,34
- Six observational studies16,19,30,35–37 evaluated the presence of myoclonus within 96 hours after arrest with specificity for poor outcome ranging from 77.8% to 97.4%. There were methodological limitations in all studies, including a lack of standard definitions, lack of blinding, incomplete data about EEG correlates, and the inability to differentiate subtypes of myoclonus. The literature was so imprecise as to make it potentially harmful if undifferentiated myoclonus is used as a prognostic marker.
- Historically, the best motor examination in the upper extremities has been used as a prognostic tool, with extensor or absent movement being correlated with poor outcome. The previous literature was limited by methodological concerns, including around inadequate control for effects of TTM and medications and self-fulfilling prophecies, and there was a lower-than-acceptable false-positive rate (10% to 15%).7 The performance of the motor examination was not evaluated in the 2020 ILCOR systematic review. The updates made to the 2015 recommendations are based on concerns that the motor examination is subject to confounding and has an unacceptably high false-positive rate and, thus, should not be used as a prognostic tool or as a screen for subsequent testing.
Use of Serum Biomarkers for Neuroprognostication
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. When performed in combination with other prognostic tests, it may be reasonable to consider high serum values of neuron-specific enolase (NSE) within 72 h after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | C-LD | 2. The usefulness of S100 calcium-binding protein (S100B), Tau, neurofilament light chain, and glial fibrillary acidic protein in neuroprognostication is uncertain |
Synopsis
Serum biomarkers are blood-based tests that measure the concentration of proteins normally found in the central nervous system (CNS). These proteins are absorbed into blood in the setting of neurological injury, and their serum levels reflect the degree of brain injury. Limitations to their prognostic utility include variability in testing methods on the basis of site and laboratory, between-laboratory inconsistency in levels, susceptibility to additional uncertainty due to hemolysis, and potential extracerebral sources of the proteins. NSE and S100B are the 2 most commonly studied markers, but others are included in this review as well. The 2020 ILCOR systematic review evaluated studies that obtained serum biomarkers within the first 7 days after arrest and correlated serum biomarker concentrations with neurological outcome. Other testing of serum biomarkers, including testing levels over serial time points after arrest, was not evaluated. A large observational cohort study investigating these and other novel serum biomarkers and their performance as prognostic biomarkers would be of high clinical significance.
Recommendation-Specific Supportive Text
- Twelve observational studies evaluated NSE collected within 72 hours after arrest.10,13,21,23,38–45 The maximal level that correlated with poor outcome ranged from 33 to 120 μg/L with specificity for poor outcome of 75% to 100%. The evidence is limited because of lack of blinding, laboratory inconsistencies, a broad range of thresholds needed to achieve 100% specificity, and imprecision. As such, an absolute value cutoff of NSE that predicts poor prognosis is not known, though very high levels of NSE may be used as part of multimodal prognostication. There is research interest in evaluating serial measures over the first days after arrest as a prognostic tool instead of using a single absolute value.10,46
- Three observational studies40,47,48 evaluated S100B levels within the first 72 hours after arrest. The maximal level that correlated with poor outcome ranged broadly depending on the study and the timing when it was measured after arrest. At values reported to achieve 100% specificity, test sensitivity ranged from 2.8% to 77.6%. The evidence is limited by the small number of studies and the broad range of thresholds across the studies required to achieve 100% specificity. The ILCOR review also evaluated 1 study each evaluating glial fibrillary acidic protein44 and Tau49 and 2 studies evaluating neurofilament light chain.50,51 Given the low number of studies, the LOE was low, and these serum biomarkers could not be recommended for clinical practice.
These recommendations are supported by the 2020 CoSTR for ALS,4 which supplements the last comprehensive review of this topic conducted in 2015.7
Use of Electrophysiological Tests for Neuroprognostication
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. When evaluated with other prognostic tests, the prognostic value of seizures in patients who remain comatose after cardiac arrest is uncertain. |
| 2b | B-NR | 2. When performed with other prognostic tests, it may be reasonable to consider persistent status epilepticus 72 h or more after cardiac arrest to support the prognosis of poor neurological outcome. |
| 2b | B-NR | 3. When performed with other prognostic tests, it may be reasonable to consider burst suppression on EEG in the absence of sedating medications at 72 h or more after arrest to support the prognosis of poor neurological outcome. |
| 2b | B-NR | 4. When performed with other prognostic tests, it may be reasonable to consider bilaterally absent N20 somatosensory evoked potential (SSEP) waves more than 24 h after cardiac arrest to support the prognosis of poor neurological outcome. |
| 2b | B-NR | 5. When evaluated with other prognostic tests after arrest, the usefulness of rhythmic periodic discharges to support the prognosis of poor neurological outcome is uncertain. |
| 3: No Benefit | B-NR | 6. We recommend that the absence of EEG reactivity within 72 h after arrest not be used alone to support a poor neurological prognosis. |
Synopsis
Electroencephalography is widely used in clinical practice to evaluate cortical brain activity and diagnose seizures. Its use as a neuroprognostic tool is promising, but the literature is limited by several factors: lack of standardized terminology and definitions, relatively small sample sizes, single center study design, lack of blinding, subjectivity in the interpretation, and lack of accounting for effects of medications. There is also inconsistency in definitions used to describe specific findings and patterns. EEG patterns that were evaluated in the 2020 ILCOR systematic review include unreactive EEG, epileptiform discharges, seizures, status epilepticus, burst suppression, and “highly malignant” EEG. Unfortunately, different studies define highly malignant EEG differently or imprecisely, making use of this finding unhelpful.
SSEPs are obtained by stimulating the median nerve and evaluating for the presence of a cortical N20 wave. Bilaterally absent N20 SSEP waves have been correlated with poor prognosis, but reliability of this modality is limited by requiring appropriate operator skills and care to avoid electric interference from muscle artifacts or from the ICU environment. One benefit to SSEPs is that they are subject to less interference from medications than are other modalities.
Recommendation-Specific Supportive Text
- Five observational studies35,52–55 evaluated the role of electrographic and/or convulsive seizures in neuroprognostication. The studies focused on electrographic seizures, though some studies also included convulsive seizures. Although the specificity of seizures in the studies included in the ILCOR systematic review was 100%, sensitivity of this finding was poor (0.6% to 26.8%), and other studies that were not included in the review found patients with postarrest seizures who had good outcomes.36,56,57 Additional methodological concerns include selection bias for which patients underwent EEG monitoring and inconsistent definitions of seizure. The term seizure encompasses a broad spectrum of pathologies that likely have different prognoses, ranging from a single brief electrographic seizure to refractory status epilepticus, and this imprecision justified the more limited recommendation.
- Six observational studies21,55,58–61 evaluated status epilepticus within 5 days after arrest and evaluated outcomes at time points ranging from hospital discharge to 6 months after arrest. The specificity of status epilepticus for poor outcome ranged from 82.6% to 100%. Interestingly, although status epilepticus is a severe form of seizures, the specificity of status epilepticus for poor outcome was less than that which was reported in the studies examining the seizures overall (as above). Additional concerns include the inconsistent definition of status epilepticus, lack of blinding, and the use of status epilepticus to justify withdrawal of life-sustaining therapies leading to potential self-fulfilling prophecies.
- Six studies21,35,54,59,62,63 evaluated burst suppression within 120 hours after arrest. One additional study64 subdivided burst suppression into synchronous versus heterogeneous patterns. Definitions of burst suppression varied or were not specified. Specificity ranged from 90.7% to 100%, and sensitivity was 1.1% to 51%. The lack of standardized definitions, potential for self-fulfilling prophecies, and the lack of controlling for medication effects limited the ability to make a stronger recommendation, despite the overall high specificity. Additional focus on identifying subtypes of burst suppression, such as the synchronous subtype (which appeared to be highly specific in a single study), should be investigated further. Burst suppression can be caused by medications, so it is particularly important that providers have knowledge about the potential effects of medication on this prognostic tool.
- Fourteen observational studies9,13,15–17,23,59,64–70 evaluated bilaterally absent N20 SSEP waves within 96 hours after arrest and correlated the finding with outcome at time points ranging from hospital discharge to 6 months after arrest. Specificity ranged from 50% to 100%. Three studies had specificity below 100%, and additional methodological limitations included lack of blinding and potential for self-fulfilling prophecies. While the studies evaluated SSEPs obtained at any time starting immediately after arrest, there is a high likelihood of potential confounding factors early after arrest, leading to the recommendation that SSEPs should only be obtained more than 24 hours after arrest.
- Discharges on EEG were divided into 2 types: rhythmic/periodic and nonrhythmic/periodic. Nine observational studies evaluated rhythmic/ periodic discharges.16,45,52–54,61,63,66,69 The specificity of rhythmic/periodic discharges ranged from 66.7% to 100%, with poor sensitivity (2.4%–50.8%). The studies evaluating rhythmic/periodic discharges were inconsistent in the definitions of discharges. Most did not account for effects of medications, and some studies found unacceptably low specificity. Nonetheless, as the time from the cardiac arrest increased, the specificity of rhythmic/periodic discharges for poor outcome improved. There is opportunity to develop this EEG finding as a prognostic tool. Five observational studies52,53,64,66,69 evaluated nonrhythmic/periodic discharges. Specificity for poor outcome was low over the entire post–cardiac arrest period evaluated in the studies.
- Ten observational studies16,30,53–55,62,65,71–73 reported on the prognostic value of unreactive EEG. Specificity ranged from 41.7% to 100% and was below 90% in most studies. There was inconsistency in the definitions of and stimuli used for EEG reactivity. Studies also did not account for effects of temperature and medications. Thus, the overall certainty of the evidence was rated as very low.
These recommendations are supported by the 2020 CoSTR for ALS,4 which supplements the last comprehensive review of this topic conducted in 2015.7
Use of Neuroimaging for Neuroprognostication
| COR | LOE | Recommendations |
|---|---|---|
| 2b | B-NR | 1. When performed with other prognostic tests, it may be reasonable to consider reduced gray-white ratio (GWR) on brain computed tomography (CT) after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | B-NR | 2. When performed with other prognostic tests, it may be reasonable to consider extensive areas of restricted diffusion on brain MRI at 2 to 7 days after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
| 2b | B-NR | 3. When performed with other prognostic tests, it may be reasonable to consider extensive areas of reduced apparent diffusion coefficient (ADC) on brain MRI at 2 to 7 days after cardiac arrest to support the prognosis of poor neurological outcome in patients who remain comatose. |
Synopsis
Neuroimaging may be helpful after arrest to detect and quantify structural brain injury. CT and MRI are the 2 most common modalities. On CT, brain edema can be quantified as the GWR, defined as the ratio between the density (measured as Hounsfield units) of the gray matter and the white matter. Normal brain has a GWR of approximately 1.3, and this number decreases with edema. On MRI, cytotoxic injury can be measured as restricted diffusion on diffusion-weighted imaging (DWI) and can be quantified by the ADC. DWI/ADC is a sensitive measure of injury, with normal values ranging between 700 and 800×10−6 mm2 /s and values decreasing with injury. CT and MRI findings of brain injury evolve over the first several days after arrest, so the timing of the imaging study of interest is of particular importance as it relates to prognosis.
Recommendation-Specific Supportive Text
- Twelve studies23,24,31,38,66,74–79 evaluated GWR on head CT. Whole-brain GWR (GWR average) and GWR in specific regions were evaluated. The specificity was 85% to 100%, and only 1 study reported a specificity that was not 100%. Many of the studies evaluated head CTs that were obtained within the first 24 hours after arrest, though some studies included head CTs obtained up to 72 hours after arrest. There were methodological limitations, including selection bias, risk of multiple comparisons, and heterogeneity of measurement techniques, such as anatomic sites and calculation methods. Thus, a specific GWR threshold that predicts poor prognosis with 100% specificity is unknown. Additionally, the optimal timing for obtaining head CT after arrest to optimize the GWR as a prognostic tool is unknown.
- Five observational studies11,23,74,80,81 investigated DWI changes on MRI within 5 days after arrest. The studies evaluated MRI qualitatively for “high signal intensity” and “positive findings,” but the definitions of positive findings differed between studies and, in some studies, examined only specific brain regions. Specificity was 55.7% to 100%. The imprecise definition and short-term outcome in some studies led to significant uncertainty about how to use DWI MRI to predict poor prognosis. In the correct setting, a significant burden of DWI MRI findings or DWI MRI findings in specific regions of interest may be correlated with poor prognosis, but a broader recommendation could not be supported.
- Three observational studies82–84 investigated ADC on MRI within 7 days after arrest. The studies were designed to determine thresholds that achieved 100% specificity, though the ADC and brain volume thresholds needed to achieve that specificity varied broadly. While quantitative ADC measurements are a promising tool, their broad use is limited by feasibility concerns. Additionally, there are relatively few studies, and per other imaging features, there was heterogeneity of measurement techniques, including in sites and calculation methods. A specific ADC threshold that predicts poor prognosis is not known.
These recommendations are supported by the 2020 CoSTR for ALS,4 which supplements the last comprehensive review of this topic conducted in 2015.7
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend structured assessment for anxiety, depression, posttraumatic stress, and fatigue for cardiac arrest survivors and their caregivers. |
| 1 | C-LD | 2. We recommend that cardiac arrest survivors have multimodal rehabilitation assessment and treatment for physical, neurological, cardiopulmonary, and cognitive impairments before discharge from the hospital. |
| 1 | C-LD | 3. We recommend that cardiac arrest survivors and their caregivers receive comprehensive, multidisciplinary discharge planning, to include medical and rehabilitative treatment recommendations and return to activity/work expectations. |
| 2b | C-LD | 4. Debriefings and referral for follow-up for emotional support for lay rescuers, EMS providers, and hospital-based healthcare workers after a cardiac arrest event may be beneficial. |
Synopsis
Cardiac arrest survivors, like many survivors of critical illness, often experience a spectrum of physical, neurological, cognitive, emotional, or social issues, some of which may not become apparent until after hospital discharge. Survivorship after cardiac arrest is the journey through rehabilitation and recovery and highlights the far-reaching impact on patients, families, healthcare partners, and communities (Figure 11).1–3
The systems-of-care approach to cardiac arrest includes the community and healthcare response to cardiac arrest. However, with more people surviving cardiac arrest, there is a need to organize discharge planning and long-term rehabilitation care resources. Survivorship plans that address treatment, surveillance, and rehabilitation need to be provided at hospital discharge to optimize transitions of care to the outpatient setting. For many patients and families, these plans and resources may be paramount to improved quality of life after cardiac arrest. Survivorship plans help guide the patient, caregivers, and primary care providers and include a summary of the inpatient course, recommended follow-up appointments, and postdischarge recovery expectations (Figure 12).
Cardiac arrest survivors, their families, and families of nonsurvivors may be powerful advocates for community response to cardiac arrest and patient-centered outcomes. Enhancing survivorship and recovery after cardiac arrest needs to be a systematic priority, aligned with treatment recommendations for patients surviving stroke, cancer, and other critical illnesses.3–5
Recommendation-Specific Supportive Text
- Approximately one third of cardiac arrest survivors experience anxiety, depression, or posttraumatic stress.6–9 Fatigue is also common and may be due to physical, cognitive, or affective impairments. Family or caregivers may also experience significant stress and benefit from therapy.10–17
- Cognitive impairments after cardiac arrest include difficulty with memory, attention, and executive function.18–22 Physical, neurological, and cardiopulmonary impairments are also common.3 Early evaluation for cardiac rehabilitation and physical, occupational, and speech language therapy may be helpful to develop strategies to recover from, overcome, or adapt to impairments.3,23–25
- Community reintegration and return to work or other activities may be slow and depend on social support and relationships.26–29 Patients need direction about when to begin driving and when to return to intimacy.30,31
- Rescuers may experience anxiety or posttraumatic stress about providing or not providing BLS.23,32 Hospital-based care providers may also experience emotional or psychological effects of caring for a patient with cardiac arrest.34 Team debriefings may allow a review of team performance (education, quality improvement) as well as recognition of the natural stressors associated with caring for a patient near death.35
These recommendations are supported by “Sudden Cardiac Arrest Survivorship: a Scientific Statement From the AHA.”3
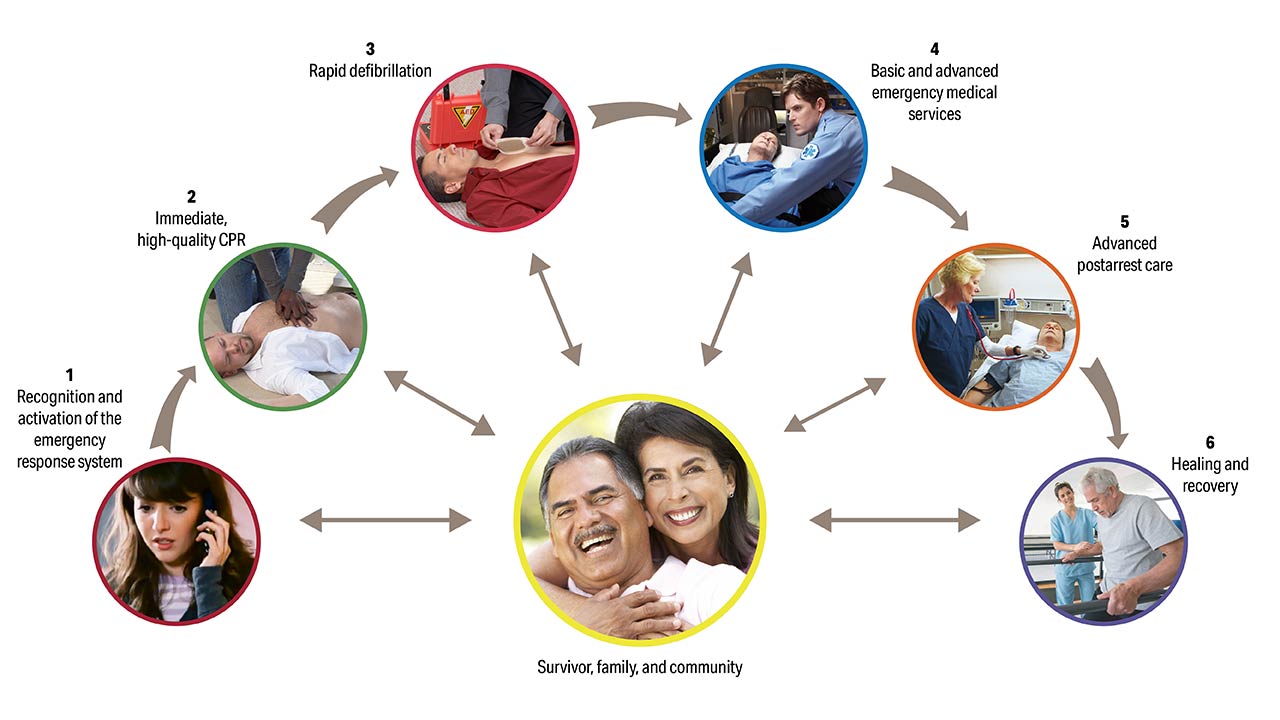
CPR indicates cardiopulmonary resuscitation.
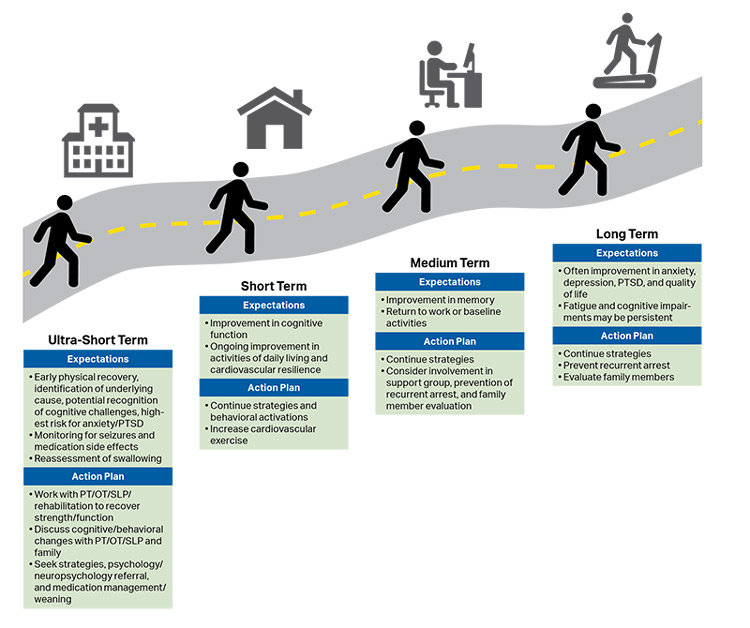
OT indicates occupational therapy; PT, physical therapy; PTSD, posttraumatic stress disorder; and SLP, speech-language pathologist
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Full resuscitative measures, including extracorporeal rewarming when available, are recommended for all victims of accidental hypothermia without characteristics that deem them unlikely to survive and without any obviously lethal traumatic injury. |
| 1 | C-EO | 2. Victims of accidental hypothermia should not be considered dead before rewarming has been provided unless there are signs of obvious death. |
| 2b | C-LD | 3. It may be reasonable to perform defibrillation attempts according to the standard BLS algorithm concurrent with rewarming strategies. |
| 2b | C-LD | 4. It may be reasonable to consider administration of epinephrine during cardiac arrest according to the standard ACLS algorithm concurrent with rewarming strategies. |
Synopsis
Severe accidental environmental hypothermia (body temperature less than 30°C [86°F]) causes marked decrease in both heart rate and respiratory rate and may make it difficult to determine if a patient is truly in cardiac arrest. A victim may also appear clinically dead because of the effects of very low body temperature. Lifesaving procedures, including standard BLS and ACLS, are therefore important to continue until a patient is rewarmed unless the victim is obviously dead (eg, rigor mortis or nonsurvivable traumatic injury). Aggressive rewarming, possibly including invasive techniques, may be required and may necessitate transport to the hospital sooner than would be done in other OHCA circumstances.1 The specific care of patients who are victims of an avalanche are not included in these guidelines but can be found elsewhere.2
Recommendation-Specific Supportive Text
- Patients with accidental hypothermia often present with marked CNS and cardiovascular depression and the appearance of death or near death, necessitating the need for prompt full resuscitative measures unless there are signs of obvious death. Along with providing standard BLS and ALS treatment, next steps include preventing additional evaporative heat loss by removing wet garments and insulating the victim from further environmental exposures. For patients with severe hypothermia (less than 30°C [86°F]) with a perfusing rhythm, core rewarming is often used. Techniques include administration of warm humidified oxygen, warm IV fluids, and intrathoracic or intraperitoneal warm-water lavage.3–5 For patients with severe hypothermia and cardiac arrest, extracorporeal rewarming allows for most rapid rewarming when available.6–11 Severe hyperkalemia and very low core temperatures may also predict resuscitation futility.12,13
- When the victim is hypothermic, pulse and respiratory rates may be slow or difficult to detect,13,14 and the ECG may even show asystole, making it important to perform lifesaving interventions until the victim is warmed and/or obviously dead. Because severe hypothermia is frequently preceded by other disorders (eg, drug overdose, alcohol use, trauma), it is advisable to look for and treat these underlying conditions while simultaneously treating hypothermia.
- The hypothermic heart may be unresponsive to cardiovascular drugs, pacemaker stimulation, and defibrillation; however, the data to support this are essentially theoretical.15 If VT or VF persists after a single shock, the value of deferring subsequent defibrillations until a target temperature is achieved is uncertain. There is no evidence to suggest a benefit from deviating from standard BLS protocol for defibrillation.
- Evidence in humans of the effect of vasopressors or other medications during cardiac arrest in the setting of hypothermia consists of case reports only.11,16,17 A systematic review of several animal studies concluded that use of vasopressors during hypothermic cardiac arrest did increase ROSC.18 No evidence was identified at the time of prior review for harm from following standard ACLS, including vasopressor medications, during hypothermic cardiac arrest.
This topic last received formal evidence review in 2010.1
Introduction
Between 1.6% and 5.1% of US adults have suffered anaphylaxis.1 Approximately 200 Americans die from anaphylaxis annually, mostly from adverse reactions to medication.2 Although anaphylaxis is a multisystem disease, life-threatening manifestations most often involve the respiratory tract (edema, bronchospasm) and/or the circulatory system (vasodilatory shock). Epinephrine is the cornerstone of treatment for anaphylaxis.3–5
| COR | LOE | Recommendation |
|---|---|---|
| 1 | C-LD | 1. In cardiac arrest secondary to anaphylaxis, standard resuscitative measures and immediate administration of epinephrine should take priority. |
Recommendation-Specific Supportive Text
- There are no RCTs evaluating alternative treatment algorithms for cardiac arrest due to anaphylaxis. Evidence is limited to case reports and extrapolations from nonfatal cases, interpretation of pathophysiology, and consensus opinion. Urgent support of airway, breathing, and circulation is essential in suspected anaphylactic reactions. Because of limited evidence, the cornerstone of management of cardiac arrest secondary to anaphylaxis is standard BLS and ACLS, including airway management and early epinephrine. There is no proven benefit from the use of antihistamines, inhaled beta agonists, and IV corticosteroids during anaphylaxis-induced cardiac arrest.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Epinephrine should be administered early by intramuscular injection (or autoinjector) to all patients with signs of a systemic allergic reaction, especially hypotension, airway swelling, or difficulty breathing. |
| 1 | C-LD | 2. The recommended dose of epinephrine in anaphylaxis is 0.2 to 0.5 mg (1:1000) intramuscularly, to be repeated every 5 to 15 min as needed. |
| 1 | C-LD | 3. In patients with anaphylactic shock, close hemodynamic monitoring is recommended. |
| 1 | C-LD | 4. Given the potential for the rapid development of oropharyngeal or laryngeal edema, immediate referral to a health professional with expertise in advanced airway placement, including surgical airway management, is recommended. |
| 2a | C-LD | 5. When an IV line is in place, it is reasonable to consider the IV route for epinephrine in anaphylactic shock, at a dose of 0.05 to 0.1 mg (0.1 mg/mL, aka 1:10 000). |
| 2a | C-LD | 6. IV infusion of epinephrine is a reasonable alternative to IV boluses for treatment of anaphylaxis in patients not in cardiac arrest. |
| 2b | C-LD | 7. IV infusion of epinephrine may be considered for post-arrest shock in patients with anaphylaxis. |
Recommendation-Specific Supportive Text
- All patients with evidence of anaphylaxis require early treatment with epinephrine. Severe anaphylaxis may cause complete obstruction of the airway and/or cardiovascular collapse from vasogenic shock. Administration of epinephrine may be lifesaving.6 Intramuscular is the preferred initial route because of ease of administration, effectiveness, and safety.7
- Injection of epinephrine into the lateral aspect of the thigh produces rapid peak plasma epinephrine concentrations.7 The adult epinephrine intramuscular autoinjector will deliver 0.3 mg of epinephrine, and the pediatric epinephrine intramuscular autoinjector will deliver 0.15 mg of epinephrine. Many patients will require additional doses, with recurrence of symptoms after 5 to 15 minutes reported.8
- Patients in anaphylactic shock are critically ill, and cardiovascular and respiratory status can change quickly, making close monitoring imperative.9
- When anaphylaxis produces obstructive airway edema, rapid advanced airway management is critical. In some cases, emergency cricothyroidotomy or tracheostomy may be required.10,11
- IV epinephrine is an appropriate alternative to intramuscular administration in anaphylactic shock when an IV is in place. An IV dose of 0.05 to 0.1 mg (5% to 10% of the epinephrine dose used routinely in cardiac arrest) has been used successfully for anaphylactic shock.9 Although not specifically studied by this route in anaphylaxis, IO epinephrine is also likely to be effective at comparable doses.
- In a canine model of anaphylactic shock, a continuous infusion of epinephrine was more effective at treating hypotension than no treatment or bolus epinephrine treatment were.12 If shock recurs after initial treatment, IV infusion (5–15 μg/min) may also better allow for careful titration and avoidance of overdosing epinephrine.
- Although data specific to patients with ROSC after cardiac arrest from anaphylaxis was not identified, an observational study of anaphylactic shock suggests that IV infusion of epinephrine (5–15 μg/min), along with other resuscitative measures such as volume resuscitation, can be successful in the treatment of anaphylactic shock.13 Because of its role in the treatment of anaphylaxis, epinephrine is a logical choice for the treatment of postarrest shock in this setting.
This topic last received formal evidence review in 2010.14
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. For asthmatic patients with cardiac arrest, sudden elevation in peak inspiratory pressures or difficulty ventilating should prompt evaluation for tension pneumothorax. |
| 2a | C-LD | 2. Due to the potential effects of intrinsic positive end-expiratory pressure (auto-PEEP) and risk of barotrauma in an asthmatic patient with cardiac arrest, a ventilation strategy of low respiratory rate and tidal volume is reasonable. |
| 2a | C-LD | 3. If increased auto-PEEP or sudden decrease in blood pressure is noted in asthmatics receiving assisted ventilation in a periarrest state, a brief disconnection from the bag mask or ventilator with compression of the chest wall to relieve air-trapping can be effective. |
Synopsis
Severe exacerbations of asthma can lead to profound respiratory distress, retention of carbon dioxide, and air trapping, resulting in acute respiratory acidosis and high intrathoracic pressure. Deaths from acute asthma have decreased in the United States, but asthma continues to be the acute cause of death for over 3500 adults per year.1,2 Patients with respiratory arrest from asthma develop life-threatening acute respiratory acidosis.3 Both the profound acidemia and the decreased venous return to the heart from elevated intrathoracic pressure are likely causes of cardiac arrest in asthma.
Care of any patient with cardiac arrest in the setting of acute exacerbation of asthma begins with standard BLS. There are also no specific alterations to ACLS for patients with cardiac arrest from asthma, although airway management and ventilation increase in importance given the likelihood of an underlying respiratory cause of arrest. Acute asthma management was reviewed in detail in the 2010 Guidelines.4 For 2020, the writing group focused attention on additional ACLS considerations specific to asthma patients in the immediate periarrest period.
Recommendation-Specific Supportive Text
- Tension pneumothorax is a rare life-threatening complication of asthma and a potentially reversible cause of arrest.5 Although usually occurring in patients receiving mechanical ventilation, cases in spontaneously breathing patients have been reported.5–7 High peak airway pressures resulting from positive-pressure ventilation can lead to pneumothorax. While difficulty ventilating an asthmatic patient in extremis is more likely due to hyperinflation and high intrathoracic pressure, evaluation for tension pneumothorax remains important.
- The acute respiratory failure that can precipitate cardiac arrest in asthma patients is characterized by severe obstruction leading to air trapping. Because of the limitation in exhalational air flow, delivery of large tidal volumes at a higher respiratory rate can lead to progressive worsening of air trapping and a decrease in effective ventilation. An approach using lower tidal volumes, lower respiratory rate, and increased expiratory time may minimize the risk of auto-PEEP and barotrauma.8
- Breath stacking in an asthma patient with limited ability to exhale can lead to increases in intrathoracic pressure, decreases in venous return and coronary perfusion pressure, and cardiac arrest.9–11 This can manifest as increased difficulty ventilating a patient, high airway pressure alarms on a ventilator, or sudden decreases in blood pressure. Brief disconnection from the ventilator or a pause in bag-mask ventilation and compression of the thorax to aid exhalation may relieve hyperinflation.
This topic last received formal evidence review in 2010.4
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. External chest compressions should be performed if emergency resternotomy is not immediately available. |
| 1 | C-LD | 2. In a trained provider-witnessed arrest of a post–cardiac surgery patient, immediate defibrillation for VF/VT should be performed. CPR should be initiated if defibrillation is not successful within 1 min. |
| 1 | C-EO | 3. In a trained provider-witnessed arrest of a post–cardiac surgery patient where pacer wires are already in place, we recommend immediate pacing in an asystolic or bradycardic arrest. CPR should be initiated if pacing is not successful within 1 min. |
| 2a | B-NR | 4. For patients with cardiac arrest after cardiac surgery, it is reasonable to perform resternotomy early in an appropriately staffed and equipped ICU. |
| 2a | C-LD | 5. Open-chest CPR can be useful if cardiac arrest develops during surgery when the chest or abdomen is already open, or in the early postoperative period after cardiothoracic surgery. |
| 2b | C-LD | 6. In post–cardiac surgery patients who are refractory to standard resuscitation procedures, mechanical circulatory support may be effective in improving outcome. |
Synopsis
Cardiac arrest occurs after 1% to 8% of cardiac surgery cases.1–8 Etiologies include tachyarrhythmias such as VT or VF, bradyarrhythmias such as heart block or asystole, obstructive causes such as tamponade or pneumothorax, technical factors such as dysfunction of a new valve, occlusion of a grafted artery, or bleeding. Like all patients with cardiac arrest, the immediate goal is restoration of perfusion with CPR, initiation of ACLS, and rapid identification and correction of the cause of cardiac arrest. Unlike most other cardiac arrests, these patients typically develop cardiac arrest in a highly monitored setting such as an ICU, with highly trained staff available to perform rescue therapies.
These guidelines are not meant to be comprehensive. A recent consensus statement on this topic has been published by the Society of Thoracic Surgeons.9
Recommendation-Specific Supportive Text
- Case reports have rarely described damage to the heart due to external chest compressions.10–14 However, other case series have not reported such damage,8 and external chest compressions remain the only means of providing perfusion in some circumstances. In this case, the risk of external chest compressions is far outweighed by the certain death in the absence of perfusion.
- VF is the presenting rhythm in 25% to 50% of cases of cardiac arrest after cardiac surgery. Immediate defibrillation by a trained provider presents distinct advantages in these patients, whereas the morbidity associated with external chest compressions or resternotomy may substantially impact recovery. Sparse data have been published addressing this question. Limited data are available from defibrillator threshold testing with backup transthoracic defibrillation, using variable waveforms and energy doses.15–17 First shock success over 90% was observed in most of these studies, though pooled results from 15 studies found a defibrillation success rate of 78% for the first shock, 35% for the second, and 14% for the third shock.18 The Society of Thoracic Surgeons Task Force on Resuscitation After Cardiac Surgery9 and the European Association for Cardio-Thoracic Surgery18 recommend 3 stacked defibrillations within 1 minute, before initiation of CPR. This departure from standard ACLS is likely warranted in the post–cardiac surgery setting because of the highly monitored setting and unique risks of compressions and resternotomy.
- In post–cardiac surgery patients with asystole or bradycardic arrest in the ICU with pacing leads in place, pacing can be initiated immediately by trained providers. Available hemodynamic monitoring modalities in conjunction with manual pulse detection provide an opportunity to confirm myocardial capture and adequate cardiac function. When pacing attempts are not immediately successful, standard ACLS including CPR is indicated. This protocol is supported by the surgical societies,9,18 though no data are available to support its use.
- No RCTs of resternotomy timing have been performed. However, good outcomes have been observed with rapid resternotomy protocols when performed by experienced providers in an appropriately equipped ICU.1,4,8,19–25 Other studies are neutral or show no benefit of resternotomy compared with standard therapy.3,6,26,27 Resternotomy performed outside of the ICU results in poor outcomes.1,3 The Society of Thoracic Surgeons recommends that resternotomy be a standard part of the resuscitation protocols for at least 10 days after surgery.9
- No randomized RCTs have been performed comparing open-chest with external CPR. Two small studies have demonstrated improved hemodynamic effects of open-chest CPR when compared with external chest compressions in cardiac surgery patients.3,4
- Multiple case series have demonstrated potential benefit from mechanical circulatory support including ECMO and cardiopulmonary bypass in patients who are refractory to standard resuscitation procedures.24,28–34 No RCT has been performed to date.
This topic last received formal evidence review in 2010.35 These recommendations were supplemented by a 2017 review published by the Society of Thoracic Surgeons.9
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Rescuers should provide CPR, including rescue breathing, as soon as an unresponsive submersion victim is removed from the water. |
| 1 | C-LD | 2. All victims of drowning who require any form of resuscitation (including rescue breathing alone) should be transported to the hospital for evaluation and monitoring, even if they appear to be alert and demonstrate effective cardiorespiratory function at the scene. |
| 2b | C-LD | 3. Mouth-to-mouth ventilation in the water may be helpful when administered by a trained rescuer if it does not compromise safety. |
| 3: No Benefit | B-NR | 4. Routine stabilization of the cervical spine in the absence of circumstances that suggest a spinal injury is not recommended. |
Synopsis
Each year, drowning is responsible for approximately 0.7% of deaths worldwide, or more than 500 000 deaths per year.1,2 A recent study using data from the United States reported a survival rate of 13% after cardiac arrest associated with drowning.3 People at increased risk for drowning include children, those with seizure disorders, and those intoxicated with alcohol or other drugs.1 Although survival is uncommon after prolonged submersion, successful resuscitations have been reported.4–9 For this reason, scene resuscitation should be initiated and the victim transported to the hospital unless there are obvious signs of death. Standard BLS and ACLS are the cornerstones of treatment, with airway management and ventilation being of particular importance because of the respiratory cause of arrest. The evidence for these recommendations was last reviewed thoroughly in 2010.
Recommendation-Specific Supportive Text
- The duration and severity of hypoxia sustained as a result of drowning is the single most important determinant of outcome.10,11 With outcome in mind, as soon as an unresponsive submersion victim is removed from the water, rescuers should provide CPR, with rescue breathing, if appropriately trained. Prompt initiation of rescue breathing increases the victim’s chance of survival.12
- Multiple observational evaluations, primarily in pediatric patients, have demonstrated that decompensation after fresh or salt-water drowning can occur in the first 4 to 6 hours after the event.13,14 This supports transporting all victims to a medical facility for monitoring for at least 4 to 6 hours if feasible.
- The immediate cause of death in drowning is hypoxemia. Based on the training of the rescuers, and only if scene safety can be maintained for the rescuer, sometimes ventilation can be provided in the water (“in-water resuscitation”), which may lead to improved patient outcomes compared with delaying ventilation until the victim is out of the water.8
- The reported incidence of cervical spine injury in drowning victims is low (0.009%).15,16 Routine stabilization of the cervical spine in the absence of circumstances that suggest a spinal injury is unlikely to benefit the patient and may delay needed resuscitation.16,17
These recommendations incorporate the results of a 2020 ILCOR CoSTR, which focused on prognostic factors in drowning.18 Otherwise, this topic last received formal evidence review in 2010.19 These guidelines were supplemented by “Wilderness Medical Society. Clinical Practice Guidelines for the Treatment and Prevention of Drowning: 2019 Update.”20
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. For cardiac arrest with known or suspected hyperkalemia, in addition to standard ACLS care, IV calcium should be administered. |
| 1 | C-LD | 2. For cardiotoxicity and cardiac arrest from severe hypomagnesemia, in addition to standard ACLS care, IV magnesium is recommended. |
| 2b | C-EO | 3. For cardiac arrest with known or suspected hypermagnesemia, in addition to standard ACLS care, it may be reasonable to administer empirical IV calcium. |
| 3: Harm | C-LD | 4. IV bolus administration of potassium for cardiac arrest in suspected hypokalemia is not recommended. |
Synopsis
Electrolyte abnormalities may cause or contribute to cardiac arrest, hinder resuscitative efforts, and affect hemodynamic recovery after cardiac arrest. In addition to standard ACLS, specific interventions may be lifesaving for cases of hyperkalemia and hypermagnesemia.
Hyperkalemia is commonly caused by renal failure and can precipitate cardiac arrhythmias and cardiac arrest. The clinical signs associated with severe hyperkalemia (more than 6.5 mmol/L) include flaccid paralysis, paresthesia, depressed deep tendon reflexes, or shortness of breath.1–3 The early electrocardiographic signs include peaked T waves on the ECG followed by flattened or absent T waves, prolonged PR interval, widened QRS complex, deepened S waves, and merging of S and T waves.4,5 As hyperkalemia progresses, the ECG can develop idioventricular rhythms, form a sine-wave pattern, and develop into an asystolic cardiac arrest.4,5 Severe hypokalemia is less common but can occur in the setting of gastrointestinal or renal losses and can lead to life-threatening ventricular arrhythmias.6–8 Severe hypermagnesemia is most likely to occur in the obstetric setting in patients being treated with IV magnesium for preeclampsia or eclampsia. At very elevated levels, hypermagnesemia can lead to altered consciousness, bradycardia or ventricular arrhythmias, and cardiac arrest.9,10 Hypomagnesemia can occur in the setting of gastrointestinal illness or malnutrition, among other causes, and, when significant, can lead to both atrial and ventricular arrhythmias.11
Recommendation-Specific Supportive Text
- In addition to standard ACLS, several therapies have long been recommended to treat life-threatening hyperkalemia.12 These include IV administration of calcium and/or bicarbonate, insulin with glucose, and/or inhaled albuterol. Parenteral calcium may stabilize the myocardial cell membrane and is therefore the most likely to be useful during cardiac arrest and can be given by the IV or IO route. A typical dose is 5 to 10 mL of 10% calcium chloride solution, or 15 to 30 mL of 10% calcium gluconate solution, administered via IV or IO line over 2 to 5 minutes.12 Standard use of sodium polystyrene (Kayexalate) is now discouraged because of poor efficacy and the risk of bowel complications. Emergent hemodialysis in the hospital setting remains a definitive treatment for life-threatening hyperkalemia.
- Although the administration of IV magnesium has not been found to be beneficial for VF/VT in the absence of prolonged QT, consideration of its use for cardiac arrest in patients with prolonged QT is advised.13 Hypomagnesemia can cause or aggravate prolonged QT, is associated with multiple arrhythmias, and may precipitate cardiac arrest.11 This provides physiological rationale for the restoration of normal levels, although standard ACLS remains the cornerstone of treatment. Recommendations for treatment of torsades de pointes are provided in the Wide Complex Tachycardia section.
- Administration of IV or IO calcium, in the doses suggested for hyperkalemia, may improve hemodynamics in severe magnesium toxicity, supporting its use in cardiac arrest although direct evidence is lacking.14
- The controlled administration of IV potassium for ventricular arrhythmias due to severe hypokalemia may be useful, but case reports have generally included infusion of potassium and not bolus dosing.15 Bolus dosing without adverse cardiac effects was reported in at least 1 small case series of cardiac surgery patients where it was administered in a highly monitored setting by an anesthesiologist, but the efficacy of this for cardiac arrest is not known, and safety concerns remain.16
This topic last received formal evidence review in 2010.12
Introduction
Approximately 1 in 12 000 admissions for delivery in the United States results in a maternal cardiac arrest.1 Although it remains a rare event, the incidence has been increasing.2 Reported maternal and fetal/neonatal survival rates vary widely.3–8 Invariably, the best outcomes for both mother and fetus are through successful maternal resuscitation. Common causes of maternal cardiac arrest are hemorrhage, heart failure, amniotic fluid embolism, sepsis, aspiration pneumonitis, venous thromboembolism, preeclampsia/eclampsia, and complications of anesthesia.1,4,6
Current literature is largely observational, and some treatment decisions are based primarily on the physiology of pregnancy and extrapolations from nonarrest pregnancy states.9 High-quality resuscitative and therapeutic interventions that target the most likely cause of cardiac arrest are paramount in this population. Perimortem cesarean delivery (PMCD) at or greater than 20 weeks uterine size, sometimes referred to as resuscitative hysterotomy, appears to improve outcomes of maternal cardiac arrest when resuscitation does not rapidly result in ROSC (Figure 15).10–14 Further, shorter time intervals from arrest to delivery appear to lead to improved maternal and neonatal outcomes.15 However, the clinical decision to perform PMCD―and its timing with respect to maternal cardiac arrest―is complex because of the variability in level of practitioner and team training, patient factors (eg, etiology of arrest, gestational age), and system resources. Finally, case reports and case series using ECMO in maternal cardiac arrest patients report good maternal survival.16 The treatment of cardiac arrest in late pregnancy represents a major scientific gap.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Team planning for cardiac arrest in pregnancy should be done in collaboration with the obstetric, neonatal, emergency, anesthesiology, intensive care, and cardiac arrest services. |
| 1 | C-LD | 2. Because immediate ROSC cannot always be achieved, local resources for a perimortem cesarean delivery should be summoned as soon as cardiac arrest in a woman in the second half of pregnancy is recognized. |
| 1 | C-EO | 3. Protocols for management of OHCA in pregnancy should be developed to facilitate timely transport to a center with capacity to immediately perform perimortem cesarean delivery while providing ongoing resuscitation. |
Recommendation-Specific Supportive Text
- To assure successful maternal resuscitation, all potential stakeholders need to be engaged in the planning and training for cardiac arrest in pregnancy, including the possible need for PMCD. Based on similarly rare but time-critical interventions, planning, simulation training and mock emergencies will assist in facility preparedness.17–21
- Since initial efforts for maternal resuscitation may not be successful, preparation for PMCD should begin early in the resuscitation, since decreased time to PMCD is associated with better maternal and fetal outcomes.8
- In cases of prehospital maternal arrest, rapid transport directly to a facility capable of PMCD and neonatal resuscitation, with early activation of the receiving facility’s adult resuscitation, obstetric, and neonatal resuscitation teams, provides the best chance for a successful outcome.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. Priorities for the pregnant woman in cardiac arrest should include provision of high-quality CPR and relief of aortocaval compression through left lateral uterine displacement. |
| 1 | C-LD | 2. Because pregnant patients are more prone to hypoxia, oxygenation and airway management should be prioritized during resuscitation from cardiac arrest in pregnancy. |
| 1 | C-EO | 3. Because of potential interference with maternal resuscitation, fetal monitoring should not be undertaken during cardiac arrest in pregnancy. |
| 1 | C-EO | 4. We recommend targeted temperature management for pregnant women who remain comatose after resuscitation from cardiac arrest. |
| 1 | C-EO | 5. During targeted temperature management of the pregnant patient, it is recommended that the fetus be continuously monitored for bradycardia as a potential complication, and obstetric and neonatal consultation should be sought. |
Recommendation-Specific Supportive Text
- The gravid uterus can compress the inferior vena cava, impeding venous return, thereby reducing stroke volume and cardiac output. In the supine position, aortocaval compression can occur for singleton pregnancies starting at approximately 20 weeks of gestational age or when the fundal height is at or above the level of the umbilicus.22 Manual left lateral uterine displacement effectively relieves aortocaval pressure in patients with hypotension (Figure 16).23,23a,23b
- Airway, ventilation, and oxygenation are particularly important in the setting of pregnancy because of increased maternal metabolism and decreased functional reserve capacity due to the gravid uterus, making pregnant patients more prone to hypoxia. Furthermore, fetal hypoxia has known detrimental effects. Both of these considerations support earlier advanced airway management for the pregnant patient.
- Resuscitation of the pregnant woman, including PMCD when indicated, is the first priority because it may lead to increased survival of both the woman and the fetus.9 Fetal monitoring does not achieve this goal and may distract from maternal resuscitation efforts, particularly defibrillation and preparation of the abdomen for PMCD.
- There are no randomized trials of the use of TTM in pregnancy. However, there are several case reports of good maternal and fetal outcome with the use of TTM after cardiac arrest.24,25
- After successful maternal resuscitation, the undelivered fetus remains susceptible to the effects of hypothermia, acidosis, hypoxemia, and hypotension, all of which can occur in the setting of post-ROSC care with TTM. In addition, deterioration of fetal status may be an early warning sign of maternal decompensation.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. During cardiac arrest, if the pregnant woman with a fundus height at or above the umbilicus has not achieved ROSC with usual resuscitation measures plus manual left lateral uterine displacement, it is advisable to prepare to evacuate the uterus while resuscitation continues. |
| 1 | C-LD | 2. In situations such as nonsurvivable maternal trauma or prolonged pulselessness, in which maternal resuscitative efforts are considered futile, there is no reason to delay performing perimortem cesarean delivery in appropriate patients. |
| 2a | C-EO | 3. To accomplish delivery early, ideally within 5 min after the time of arrest, it is reasonable to immediately prepare for perimortem cesarean delivery while initial BLS and ACLS interventions are being performed. |
Recommendation-Specific Supportive Text
- Evacuation of the gravid uterus relieves aortocaval compression and may increase the likelihood of ROSC.10–14 In the latter half of pregnancy, PMCD may be considered part of maternal resuscitation, regardless of fetal viability.26
- Early delivery is associated with better maternal and neonatal survival.15 In situations incompatible with maternal survival, early delivery of the fetus may also improve neonatal survival.26
- The optimal timing for the performance of PMCD is not well established and must logically vary on the basis of provider skill set and available resources as well as patient and/or cardiac arrest characteristics. A systematic review of the literature evaluated all case reports of cardiac arrest in pregnancy about the timing of PMCD, but the wide range of case heterogeneity and reporting bias does not allow for conclusions.15 Survival of the mother has been reported up to 39 minutes after the onset of maternal cardiac arrest.4,10,27–29 In a systematic review of literature published 1980 to 2010, the median time from maternal cardiac arrest to delivery was 9 minutes in surviving mothers and 20 minutes in nonsurviving mothers.15 In the same study, the median time to PMCD was 10 minutes in surviving and 20 minutes in nonsurviving neonates. The time to delivery was within 4 minutes in only 4/57 (7%) reported cases.15 In a UK cohort study,4 the median time from collapse to PMCD was 3 minutes in women who survived compared with 12 minutes in nonsurvivors. In this study, 24/25 infants survived when PMCD occurred within 5 minutes after maternal cardiac arrest compared with 7/10 infants when PMCD occurred more than 5 minutes after cardiac arrest. Neonatal survival has been documented with PMCD performed up to 30 minutes after the onset of maternal cardiac arrest.10 The expert recommendation for timing for PMCD in cardiac arrest at less than 5 minutes remains an important goal, though rarely achieved.9 There is no evidence for a specific survival threshold at 4 minutes.8
These recommendations are supported by “Cardiac Arrest in Pregnancy: a Scientific Statement From the AHA”9 and a 2020 evidence update.30
| COR | LOE | Recommendations |
|---|---|---|
| 2a | C-LD | 1. In patients with confirmed pulmonary embolism as the precipitant of cardiac arrest, thrombolysis, surgical embolectomy, and mechanical embolectomy are reasonable emergency treatment options. |
| 2b | C-LD | 2. Thrombolysis may be considered when cardiac arrest is suspected to be caused by pulmonary embolism. |
Synopsis
This topic was reviewed in an ILCOR systematic review for 2020.1 PE is a potentially reversible cause of shock and cardiac arrest. Acute increase in right ventricular pressure due to pulmonary artery obstruction and release of vasoactive mediators produces cardiogenic shock that may rapidly progress to cardiovascular collapse. Management of acute PE is determined by disease severity.2 Fulminant PE, characterized by cardiac arrest or severe hemodynamic instability, defines the subset of massive PE that is the focus of these recommendations. Pulseless electrical activity is the presenting rhythm in 36% to 53% of PE-related cardiac arrests, while primary shockable rhythms are uncommon.3–5
Prompt systemic anticoagulation is generally indicated for patients with massive and submassive PE to prevent clot propagation and support endogenous clot dissolution over weeks. Anticoagulation alone is inadequate for patients with fulminant PE. Pharmacological and mechanical therapies to rapidly reverse pulmonary artery occlusion and restore adequate pulmonary and systemic circulation have emerged as primary therapies for massive PE, including fulminant PE.2,6 Current advanced treatment options include systemic thrombolysis, surgical or percutaneous mechanical embolectomy, and ECPR.
Recommendation-Specific Supportive Text
- In the 2020 ILCOR systematic review, no randomized trials were identified addressing the treatment of cardiac arrest caused by confirmed PE. Observational studies of fibrinolytic therapy for suspected PE were found to have substantial bias and showed mixed results in terms of improvement in outcomes.3,7–10 Two case series totaling 21 patients with PE undergoing CPR who underwent surgical embolectomy reported 30-day survival rates of 12.5% and 71.4%, respectively.11,12 A case series of patients with PE-related cardiac arrest reported ROSC in 6 of 7 patients (86%) treated with percutaneous mechanical thrombectomy.13 In terms of potential adverse effects, a clinical trial and several observational studies show that the risk of major bleeding in patients receiving thrombolysis and CPR is relatively low.7–9 In spite of the uncertainty of benefit, the risk of death from cardiac arrest outweighs the risk of bleeding from thrombolysis and/or the risks of mechanical or surgical interventions. Because there is no clear benefit to one approach over the other, choice of thrombolysis or surgical or mechanical thrombectomy will depend on timing and available expertise.
- The approach to cardiac arrest when PE is suspected but not confirmed is less clear, given that a misdiagnosis could place the patient at risk for bleeding without benefit. Recent evidence, however, suggests that the risk of major bleeding is not significantly higher in cardiac arrest patients receiving thrombolysis.8 PE is difficult to diagnose in the intra-arrest setting, and when ROSC is not obtained and PE is strongly suspected, the evidence supports consideration of thrombolysis.1
These recommendations are supported by a 2020 ILCOR systematic review.1
Introduction
Benzodiazepines are commonly used sedative-hypnotics used to treat anxiety, insomnia, seizures, and withdrawal and as a component of general anesthesia and procedural sedation. Benzodiazepines are implicated in a large number of poisoning-related deaths, usually in combination with other CNS depressants, such as opioids or alcohol.1
Benzodiazepine overdose causes CNS depression through agonist effects at the gamma-amino butyric acid-A receptor with resultant respiratory compromise through loss of protective airway reflexes. The subsequent hypoxemia and hypercarbia cause tissue injury and death. Patients with benzodiazepine poisoning can be readily managed with standard life support measures. Immediate treatment includes establishing an open airway and providing bag-mask ventilation, followed by endotracheal intubation when appropriate.
Flumazenil, a competitive antagonist at the gamma-amino butyric acid-A/benzodiazepine receptor, reverses CNS and respiratory depression, potentially preventing the need for intubation and mechanical ventilation. However, flumazenil administration may precipitate refractory benzodiazepine withdrawal and seizures in patients with benzodiazepine tolerance.2 Flumazenil-provoked seizures are reported in patients with preexisting seizure disorder, even in the absence of other risk factors.3 Flumazenil removes benzodiazepine-mediated suppression of sympathetic tone and may precipitate dysrhythmias, including supraventricular tachycardia, ventricular dysrhythmias, and asystole, particularly in the presence of prodysrhythmic drugs (such as cyclic antidepressants) or hypoxia.2, 4-7 Flumazenil may not fully reverse respiratory depression, particularly in mixed overdoses.8
Overdose with multiple drugs is common. Benzodiazepine overdose should not preclude the timely administration of naloxone when opioid overdose is suspected. This is particularly important given the presence of opioid-adulterated illicit drugs.
| COR | LOE | Recommendation |
|---|---|---|
| 2a | B-NR | 1. It is reasonable to administer naloxone first (before other antidotes) for respiratory depression/respiratory arrest if combined opioid and benzodiazepine poisoning is suspected. |
| 2a | B-NR | 2. Flumazenil can be effective in select patients with respiratory depression/respiratory arrest caused by pure benzodiazepine poisoning who do not have contraindications to flumazenil. |
| 3: No Benefit | C-EO | 3. Flumazenil has no role in cardiac arrest related to benzodiazepine poisoning. |
| 3: Harm | B-R | 4. Flumazenil administration is associated with harm in patients who are at increased risk for seizures or dysrhythmias. |
Recommendation-Specific Supportive Text
- Isolated benzodiazepine poisoning rarely causes life-threatening hypoventilation or hemodynamic instability.1, 9Consider the presence of concomitant opioid, ethanol, or other CNS depressant poisoning in these presentations. Opioid poisoning is more common and causes more significant respiratory depression than benzodiazepine poisoning, and naloxone has a better safety profile than flumazenil.
- Flumazenil is safe in some low-risk presentations (eg, pediatric exploratory ingestions and iatrogenic overdoses during procedural sedation) and when high-risk conditions (eg, chronic benzodiazepine dependence and coingestion of other dangerous substances) can be reliably excluded.10
- Flumazenil does not directly affect cardiac rhythm or restore spontaneous circulation.
- In a meta-analysis of randomized clinical trials in patients with presumed benzodiazepine overdose, higher rates of serious adverse effects, including seizures and dysrhythmias, occurred with flumazenil compared with standard care alone.2 Harms were uncommon and, in most cases, readily managed. The risks of flumazenil likely exceed the benefit in patients with undifferentiated coma for whom past medical history, substance use history, and the potential poison(s) involved are unknown.
Introduction
β-Adrenergic receptor antagonists (commonly called beta blockers [BBs]) are a leading cause of poisoning mortality.1 Patients with severe BB poisoning develop hypotension due to bradycardia and reduced cardiac contractility.2
Some BBs also cause dysrhythmias from sodium and/or potassium channel blockade. Bradycardia is due to direct effects on the β1-andrenergic receptor. Hypotension, which can be cardiogenic, vasodilatory from α1-andrenergic receptor antagonism, or multifactorial, is often refractory to vasopressor therapy. BBs are sometimes associated with hypoglycemia,3, 4 though this relationship is complex.5, 6
Commonly employed treatment modalities include atropine, glucagon, calcium, vasopressors, high-dose insulin, and ILE therapy. In some refractory cases, VA-ECMO has been employed. No studies have evaluated the use of these therapies for cardiac arrest due to BB poisoning. Therefore, recommendations are derived from studies in poisoned patients with severe BB-induced shock.
The treatment of patients with life-threatening sodium channel blockade due to severe poisoning is discussed in section 13 of this focused update, and specific recommendations about the use of VA-ECMO for critical poisoning are provided in section 15. Recommendations about the management of patients with long-QT syndromes and torsade de pointes were last updated by the AHA in 2020.7
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend that high-dose insulin be administered for hypotension due to beta blocker (BB) poisoning refractory to or in conjunction with vasopressor therapy. |
| 1 | C-LD | 2. We recommend that vasopressors be administered for hypotension due to BB poisoning. |
| 2a | C-LD | 3. It is reasonable to use a bolus of glucagon, followed by a continuous infusion, for bradycardia or hypotension due to BB poisoning. |
| 2a | C-LD | 4. It is reasonable to utilize extracorporeal life support techniques such as venoarterial extracorporeal membrane oxygenation (VA-ECMO) for life-threatening BB poisoning with cardiogenic shock refractory to pharmacological interventions. |
| 2b | C-LD | 5. It may be reasonable to administer atropine for BB-induced bradycardia. |
| 2b | C-LD | 6. It may be reasonable to attempt electrical pacing for BB-induced bradycardia. |
| 2b | C-LD | 7. It may be reasonable to use hemodialysis for life-threatening atenolol or sotalol poisoning. |
| 3: No Benefit | C-LD | 8. Intravenous lipid emulsion therapy is not likely to be beneficial for life-threatening BB poisoning. |
Recommendation-Specific Supportive Text
- High-dose insulin improves inotropy in cardiogenic shock from BB poisoning.8, 9 One large cohort study reports favorable outcomes with lower rates of vasoconstrictive complications than vasopressor-only therapy.10 In some cases, insulin therapy appears to be vasopressor-sparing, with recurrence of vasopressor-resistant hypotension after insulin therapy was reduced or stopped,8, 11 though this is not reported consistently.12 Protocolized care reduces the risk of hypoglycemia.13 Hypokalemia and volume overload are additional concerns.9, 10
- Successful use of inotropes and vasopressors is described in a recent systematic review of case reports, case series, and animal studies.12 Because they are readily available and act quickly, vasopressors are almost always the initial therapy for BB-induced hypotension.
- Intravenous glucagon increased contractility and improvement in hemodynamics in case reports14, 15 and case series.12 The doses used are higher than those used to treat hypoglycemia. Vomiting is common with the bolus, and rapid tachyphylaxis is described.12, 16
- On the basis of case reports,17 case series,18 and observational studies,19 VA-ECMO may be lifesaving for patients with persistent cardiogenic shock (pump failure) refractory to maximal supportive care.
- Only case reports are available to describe the use of atropine, which was associated with improvements in heart rate and blood pressure.12
- A recent systematic review showed inconsistent response to pacemaker therapy.12 Electrical and mechanical capture are not always successful, and hypotension may persist despite mechanical capture. Attempts to optimize pharmacotherapy may improve response to external or internal pacing.20
- Observational studies in patients with critical poisoning due to atenolol or sotolol and kidney impairment reported clinical improvement after the use of hemodialysis.21 Nadolol is also considered dialysable, but clinical data are lacking.21
- The use of ILE is described in case reports and observational studies of BB poisonings.22 Adverse effects are reported, including clogging of VA-ECMO filters, pancreatitis, and sudden cardiovascular collapse when ILE was administered to patients with oral BB overdose.23-25 A retrospective study did not find a benefit from ILE therapy.26 Existing evidence-based recommendations advise against the routine use of ILE for BB poisoning.27
Introduction
Antagonists of the L-type calcium channel (commonly called calcium channel blockers or CCBs) are divided into 2 pharmacologic classes: dihydropyridines (eg, nifedipine, amlodipine) and nondihydropyridines (eg, diltiazem, verapamil). At therapeutic doses, non-dihydropyridines have more pronounced effects on cardiac tissue, including the sinoatrial and atrioventricular nodes, resulting in negative chronotropy, while dihydropyridines cause peripheral vasodilation. These distinctions are often lost when therapeutic doses are exceeded and patients present with severe shock from bradycardia, vasodilation, and/or loss of inotropy. Prolonged effects are common given that CCBs are frequently prescribed in sustained-release forms (diltiazem, verapamil, nifedipine) or have very long half-lives (amlodipine). As a result, CCBs are a leading cause of poisoning mortality.1 Commonly employed treatment modalities include atropine, calcium, vasopressors, high-dose insulin therapy, nitric oxide scavengers (eg, methylene blue), and ILE therapy. Extracorporeal life support, such as VA-ECMO, can be employed in refractory cases. No randomized controlled clinical trials have evaluated the use of these therapies in the context of cardiac arrest or refractory shock. As such, recommendations are derived from lower-quality data in severely poisoned patients.
| 1 | B-NR | 1. We recommend administering vasopressors for hypotension from calcium channel blocker (CCB) poisoning. |
|---|---|---|
| 1 | B-NR | 2. We recommend administering high-dose insulin for hypotension due to CCB poisoning. |
| 2a | C-LD | 3. It is reasonable to administer calcium for CCB poisoning. |
| 2a | C-LD | 4. It is reasonable to administer atropine for hemodynamically significant bradycardia from CCB poisoning. |
| 2a | C-LD | 5. It is reasonable to utilize extracorporeal life support techniques such as VA-ECMO for cardiogenic shock due to CCB poisoning that is refractory to pharmacological interventions. |
| 2b | C-LD | 6. It might be reasonable to attempt electrical pacing for CCB poisoning with refractory bradycardia. |
| 2b | C-LD | 7. The usefulness of a glucagon bolus and infusion for CCB poisoning is uncertain. |
| 2b | C-LD | 8. The usefulness of administering methylene blue for refractory vasodilatory shock due to CCB poisoning is uncertain. |
| 3: No Benefit | C-LD | 9. The routine use of intravenous lipid emulsion (ILE) therapy for CCB poisoning is not recommended. |
Recommendation-Specific Supportive Text
- Many patients with CCB-induced shock receive a vasopressor therapy.1-3 One large retrospective case series demonstrated excellent survival rates with the primary use of vasopressors (most commonly norepinephrine, at doses up to 100 mcg/min in adults), with low rates of ischemic complications.3 Three patients in this series suffered cardiac arrest prior to vasopressor therapy. There is no evidence to guide the choice of vasopressors.
- High-dose insulin administration improves inotropy in patients with severe cardiogenic shock from CCB poisoning.4-7 One large case series reports favorable outcomes with lower rates of vasoconstrictive complications than vasopressor-only therapy.4 Survival is reported even following cardiac arrest.4, 8 Protocolized care reduces the risk of hypoglycemia.7 Hypokalemia and volume overload are additional concerns.
- The available literature on calcium monotherapy for severe CCB toxicity is limited and subject to publication bias. Improvements in heart rate, blood pressure, and conduction abnormalities are reported9, 10; however, most patients require additional treatments.3, 4, 9, 11 In 1 case series, high doses of calcium gluconate (targeting ionized calcium concentrations up to twice normal) appeared to be more effective than lower doses.9
- Atropine is commonly used as a first-line therapy for patients with bradycardia, including those with CCB poisoning.2, 4 Treatment failures are reported, but these reports are likely subject to publication bias.2, 11
- The use of VA-ECMO for patients with refractory cardiogenic shock following CCB overdose is described in case series, with reported survival rates as high as 77%.12-15 If available, VA-ECMO may be lifesaving for patients with persistent cardiogenic shock (pump failure) refractory to maximal supportive care.
- Multiple case reports describe the use of electrical pacing for patients with bradydysrhythmias and hemodynamic instability following CCB poisoning. Results are mixed.2, 3, 11, 16, 17 Electrical pacing may be reasonable for patients with hemodynamically significant bradydysrhythmias, but it is not always effective, particularly in patients with complete AV-nodal blockade and/or vasodilatory shock.2
- Glucagon is reported as an adjunctive therapy for severe CCB poisoning.3, 4, 11 Reported response rates are variable, vomiting is common, and rapid tachyphylaxis may occur.10, 18, 19
- Methylene blue, a nitric oxide scavenger, is described in case series and case reports as an effective adjunct to treat refractory vasodilatory shock following CCB overdose (primarily involving amlodipine).20 However, responses are mixed, and the effects may be transient.
- A large retrospective study did not find a benefit from ILE therapy in CCB poisoning.21 Experimental and clinical data suggest that ILE increases absorption of lipophilic drugs from the gastrointestinal tract, potentially worsening poisoning from oral overdose.8, 22 As a result, evidence-based recommendations advise against the routine use of ILE for CCB poisoning.23
Introduction
Cocaine toxicity is caused by sympathetic nervous system effects, CNS stimulation, and local anesthetic (LA) effects. Cocaine produces a sympathomimetic toxidrome marked by tachycardia, hypertension, hyperthermia, diaphoresis, increased psychomotor activity, and seizures.1 Cocaine induces tachycardia (postsynaptic β-adrenergic receptor agonism) and hypertension (peripheral postsynaptic α-adrenergic receptor agonism) by catecholamine reuptake inhibition. In addition, reuptake blockade of norepinephrine, epinephrine, dopamine, and serotonin also causes the CNS and neuropsychiatric symptoms of cocaine poisoning.1, 2
The electrocardiographic changes and dysrhythmogenic properties of cocaine are a result of the effect of cocaine on cardiac sodium and potassium channels.3 This blockade leads to slowed conductance during phase 0 of the cardiac action potential. As a result, patients develop QRS prolongation and wide-complex tachycardia (WCT) similar to what occurs with Vaughan-Williams Ia and Ic medications.4, 5 Cocaine may also cause QTc prolongation via blockade of cardiac potassium channels.1 Like other LAs, cocaine blocks neuronal sodium channels. Cocaine-induced dysrhythmias include sustained asystolic cardiac arrest and pulseless ventricular tachycardia.
Benzodiazepines remain the mainstay of initial management of blood pressure and psychomotor agitation for patients with acute cocaine poisoning. In addition, calcium channel antagonists, α1-adrenergic receptor antagonists, and nitrates can be used for severe cocaine-induced hypertension and chest pain.2, 6-8 These therapies are not germane to cardiac arrest.
| COR | LOE | Recommendation |
|---|---|---|
| 2a | C-LD | 1. It is reasonable to administer sodium bicarbonate for wide complex tachycardia or cardiac arrest from cocaine poisoning. |
| 2a | C-LD | 2. It is reasonable to administer lidocaine for wide-complex tachycardia from cocaine poisoning. |
| 2a | C-LD | 3. It is reasonable to administer vasodilators (eg, nitrates, phentolamine, calcium channel blockers) for patients with cocaine-induced coronary vasospasm. |
Recommendation-Specific Supportive Text
- Retrospective observational studies2, 9 and case reports10-13 describe the successful use of sodium bicarbonate to treat WCT from severe cocaine poisoning. A recent case report describes successful use of sodium bicarbonate in the resuscitation of a patient with asystolic cardiac arrest and subsequent wide-complex Brugada pattern.11
- Well-conducted animal studies demonstrate the ability of lidocaine to reverse cocaine-induced QRS prolongation through competitive binding between lidocaine and cocaine at the sodium channel.14, 15 Lidocaine pretreatment prevents ataxia, seizures, and death following cocaine administration in mice.10, 16 Human evidence of efficacy is limited to case reports and small retrospective studies.17, 18 Lidocaine administration has demonstrated safety in patients with cocaine-induced myocardial infarction.17 The use of lidocaine for cocaine-associated cardiac arrest is supported by case reports.18, 19
- Human clinical trials demonstrate improvements in coronary blood flow and myocardial oxygen delivery in patients with cocaine-induced coronary vasospasm after treatment with vasodilators (phentolamine, nitrates, verapamil).6, 20-23 These studies did not include patients with cardiac arrest or periarrest states. Patients with refractory ischemia from cocaine were successfully treated with phentolamine.8, 24
Introduction
Cyanide is commonly used in jewelry cleaning, electroplating, metallurgy, and other industrial and laboratory processes. Cyanide is also liberated by the in vivo metabolism of naturally occurring cyanogens (eg, linamarin and amygdalin). In structure fires, cyanide gas is liberated by the incomplete combustion of nitrogen-containing products such as plastic, vinyl, wool, and silk. Rarely, cyanide is used in criminal poisoning or suicide attempts.
Cyanide inhibits cellular respiration in the mitochondria. Patients with cyanide poisoning can rapidly develop cardiovascular collapse, metabolic acidosis with elevated plasma lactate concentrations, depressed mental status, seizures, and death.1 Confirmation of cyanide poisoning with laboratory measurement of cyanide concentrations is rarely available in clinical real time. Empiric treatment should be considered in laboratory workers, industrial workers, and victims of structure fires who present with cardiac arrest, altered mental status, elevated plasma lactate concentrations, and/or hypotension.
Hydroxocobalamin scavenges cyanide on an equimolar basis to form nontoxic cyanocobalamin. Alternatively, sodium nitrite oxidizes hemoglobin to methemoglobin, which then binds cyanide to form cyanmethemoglobin, though other mechanisms may be involved.2, 3 Sodium thiosulfate acts as a substrate for cyanide metabolism, forming thiocyanate. This process is much slower than scavenging by hydroxocobalamin and sodium nitrite. Sodium thiosulfate may work synergistically with either hydroxocobalamin or sodium nitrite. Typically, hydroxocobalamin is favored because of its rapid onset of action and simplicity of use. The principal adverse effects of hydroxocobalamin are transient hypertension and interference with colorimetric laboratory assays. Sodium nitrite administration can cause hypotension, and methemoglobin formation may worsen oxygen-carrying capacity in patients with concomitant carbon monoxide poisoning from smoke inhalation. Sodium thiosulfate, which has few adverse effects, works more slowly than the cyanide-scavenging therapies but may provide synergistic benefit, particularly when sodium nitrite is used. There are no human clinical trials comparing cyanide treatments to placebo, no human trials directly comparing cyanide treatment options alone or in combination, and no trials in human cardiac arrest.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. We recommend that hydroxocobalamin be administered for cyanide poisoning. |
| 1 | C-LD | 2. We recommend that sodium nitrite be administered for cyanide poisoning when hydroxocobalamin is unavailable or if concomitant carbon monoxide poisoning is not a concern. |
| 2a | C-LD | 3. In addition to administering hydroxocobalamin or sodium nitrite, it is reasonable to administer sodium thiosulfate for cyanide poisoning. |
| 2a | C-EO | 4. It is reasonable to administer 100% oxygen for cyanide poisoning. |
Recommendation-Specific Supportive Text
- Structure-fire victims likely represent the most common source of concern for cyanide poisoning.1, 4 Simultaneous poisoning with carbon monoxide and cyanide is common in smoke-inhalation victims. Because hydroxocobalamin does not cause hypotension or exacerbate concerns about decreased oxygen-carrying capacity, hydroxocobalamin is the primary recommended treatment for suspected cyanide poisoning.
- Sodium nitrite effectively treats cyanide poisoning and is an appropriate alternative to hydroxocobalamin, particularly when carbon monoxide poisoning is not a concern.5, 6 Dosing of sodium nitrite is challenging in children and in patients with anemia; the prescribing information lists specifications.7
- Sodium thiosulfate enhances cyanide elimination when given with hydroxocobalamin or sodium nitrite.1, 8 The mechanism of action of sodium thiosulfate is thought to be too slow to be considered monotherapy in life-threatening poisoning.
- Animal studies suggest a benefit when cyanide-specific antidotes are combined with 100% oxygen.9 There are no human studies examining the use of 100% oxygen as cyanide therapy, but it is reasonable to use 100% oxygen as therapy even with a normal partial pressure of oxygen in the context of a cellular poison, such as cyanide, that impairs cellular respiration.
Introduction
Cardiac glycoside poisoning can be caused by medications such as digoxin and digitoxin, plants such as foxglove and oleander, and certain toad venoms. Despite decreasing prescription of digoxin and digitoxin in the last decades, poisoning remains frequent because of overdosage, unintentional ingestion, drug-drug interactions, and accumulation due to reduced renal clearance. Patients with cardiac glycoside poisoning may develop gastrointestinal symptoms, confusion, hyperkalemia, and cardiac conduction abnormalities, including atrioventricular nodal block, ventricular tachycardia, ventricular fibrillation, and asystole. Although the cardiac glycosides include a range of structurally similar cardioactive steroids, most data involve digoxin poisoning.
Digoxin-specific immune antibody fragments (digoxin-fragment antigen binding [digoxin-Fab]) bind to and inactivate digoxin and structurally similar cardiac glycosides. Different dosing regimens are advocated worldwide.1-3 An observational study supports a likely survival advantage in patients who are in cardiac arrest.4 The ideal empiric dose for cardiac arrest is unknown and likely differs for digoxin poisoning compared with other cardiac glycosides.
Acute digoxin poisoning commonly causes hyperkalemia,1 and current ALS and PALS guidelines recommend administration of calcium for hyperkalemia.5, 6 Animal studies and case reports from the 1930s raised concern that calcium administration could cause cardiac arrest (“stone heart”) in this situation.7 A retrospective cohort study including mostly patients with chronic digoxin poisoning and a porcine study suggest that calcium administration is neither harmful nor beneficial.8, 9
Animal data raised concerns about defibrillation in digoxin-poisoned patients, due to the risk of precipitating new life-threatening dysrhythmias.10 Case reports in cardiac arrest11-26 showed response in some cases, absence of response in others, but no new dysrhythmia. In the absence of data to the contrary, standard ALS and PALS guidelines for defibrillation should be followed, with the addition of digoxin-Fab therapy as detailed in the following table.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend administration of digoxin-specific antibody fragments (digoxin-Fab) for digoxin or digitoxin poisoning. |
| 2a | C-LD | 2. It is reasonable to administer digoxin-Fab for poisoning due to Bufo toad venom (Chan Su) and yellow oleander. |
| 2b | C-LD | 3. It may be reasonable to consider administration of digoxin-Fab to treat poisoning from cardiac glycosides other than digoxin, Bufo toad venom, and yellow oleander. |
| 2b | C-LD | 4. It may be reasonable to administer atropine for bradydysrhythmias caused by digoxin and other cardiac glycoside poisoning. |
| 2b | C-LD | 5. It may be reasonable to attempt electrical pacing to treat bradydysrhythmias from digoxin and other cardiac glycoside poisoning. |
| 2b | C-LD | 6. It may be reasonable to administer lidocaine, phenytoin, or bretylium to treat ventricular dysrhythmias caused by digitalis and other cardiac glycoside poisoning until digoxin-Fab can be administered. |
| 3: No Benefit | B-NR | 7. We do not recommend the use of hemodialysis, hemofiltration, hemoperfusion, or plasmapheresis to treat digoxin poisoning. |
Recommendation-Specific Supportive Text
- Data from observational studies,4, 27-36 synthesized in a recent systematic review,2 show resolution of life-threatening dysrhythmias following digoxin-Fab administration. Most studies report response rates of 50% to 90%, with dysrhythmia resolution in 30 to 45 minutes in most cases. Although there are no RCTs studying cardiac arrest from digoxin or digitoxin poisoning, excellent survival (30/56 patients; 54%) was reported in an observational study of digoxin-Fab–treated patients.4 Treatment appears to be safe.37
- A randomized controlled trial among hemodynamically stable patients with yellow oleander (Thevetia peruviana, also known as Cascabela thevetia) poisoning showed promising response to digoxin-Fab,38 as do many case reports and case series.39 A Cochrane review did not identify any trials in patients with severe yellow oleander toxicity.40 In vitro studies showed affinity between bufadienolides (cardiac glycosides found in Bufo toad venom) and digoxin-Fab,41-44 murine studies showed protection,45 and some published cases showed apparent response.42, 46, 47
- Data supporting the use of digoxin-Fab to treat poisoning from cardiac glycosides other than digoxin, digitoxin, bufadienolides, and yellow oleander are limited to case reports.39, 48-54
- Published case reports describe the use of atropine to treat patients with bradycardia caused by cardiac glycoside toxicity, with variable effects.53, 55-58 No cohort studies or randomized clinical trials have been published examining atropine for digitalis toxicity.
- Two observational studies from the same center, predating the introduction of digoxin-Fab,59, 60 reported reduction in the mortality rate from 20% to 13% with transvenous pacing in digitalis-poisoned patients (mainly chronic poisoning) with bradydysrhythmias. Case reports support a role for pacing as temporizing therapy.11, 13-16, 22, 25, 26, 54, 57, 61-64 However, iatrogenic complications from transvenous pacing were reported in 36% of patients in one series.65 Some patients required a higher-than-normal current, and, in some cases, pacing could not be successfully resumed after interruption.
- Many cases in the literature report the use of antidysrhythmic medications, including lidocaine, phenytoin, or bretylium, to treat ventricular dysrhythmias caused by digoxin poisoning, with various responses.26, 57, 64, 66 However, no high-quality cohort studies or randomized trials have evaluated their effect.
- A recent systematic review found that digoxin is not well removed by extracorporeal treatments because of its large volume of distribution.67
Introduction
Local anesthetics (LAs) reversely bind sodium channels to disrupt nerve transmission and block pain signals. Patients with LA poisoning present with a constellation of CNS and cardiovascular symptoms termed local anesthetic systemic toxicity (LAST). CNS toxicity (77%–89% of patients with LAST) includes seizures (most common), agitation, syncope, dysarthria, perioral numbness, confusion, obtundation, and dizziness.1, 2 Although less common, cardiovascular toxicity (32%–55% of patients with LAST) can be life-threatening. Asystole occurred in 12% of cases, and ventricular fibrillation or ventricular tachycardia occurred in 13% of cases in 1 series.1
LAs vary in toxicity depending on the potency associated with their lipophilic side chains. Bupivacaine is a more potent cardiotoxin than ropivacaine and lidocaine in a canine model through its greater affinity and binding durations to cardiac sodium channels.3, 4 Bupivacaine may also cause reentry dysrhythmias, suppress conduction pathways, and block calcium channels.4 Optimal treatments for bupivacaine poisoning may differ from other LAs, and these differences are not well understood.4, 5
Both hypoxia and acidemia worsen toxicity from bupivacaine in animal models.5-7 Ventilation and treatment of acidemia are critical.6, 8 Many case reports of LAST occurred perioperatively, featured early advanced airway placement, and had return of spontaneous circulation through standard ALS measures without ILE.1, 2, 9 Early adjunctive administration of ILE in addition to standard ALS resuscitation is efficacious in animal models, case reports, and observational studies.5, 9-11 Other pharmacological interventions (eg, sodium bicarbonate) and mechanical support (eg, VA-ECMO) have been used for LAST, but the efficacy of these interventions remains unclear.
Evidence-based dosing recommendations for ILE are lacking. The majority of animal studies and human experience for the treatment of LAST use 20% ILE.5 Attempts to reproduce this dose using propofol (which contains 10 mg/mL propofol in 10% ILE) would likely lead to profound hypotension.
LA poisoning can also produce methemoglobinemia; treatment recommendations are provided in section 10.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. We recommend the administration of intravenous lipid emulsion for local anesthetic poisoning. |
| 1 | C-LD | 2. We recommend the use of benzodiazepines to treat seizures associated with local anesthetic systemic toxicity. |
| 2a | C-LD | 3. It is reasonable to administer sodium bicarbonate for life-threatening wide-complex tachycardia associated with local anesthetic toxicity. |
| 2a | C-EO | 4. It is reasonable to administer atropine for life-threatening bradycardia associated with local anesthetic systemic toxicity. |
| 2a | C-EO | 5. It is reasonable to utilize extracorporeal life support techniques such as VA-ECMO in local anesthetic toxicity with refractory cardiogenic shock. |
Recommendation-Specific Supportive Text
- Early administration of 20% ILE in patients with LAST is supported by animal studies, case reports, registry studies, and 1 small RCT.5, 10, 12 In conjunction with prevention of hypoxia and acidemia through standard ALS measures, administration of ILE has led to successful resuscitation in these studies. However, most of the studies are uncontrolled and subject to publication bias. The single RCT (n=16) evaluated the pharmacology and tolerability of ropivacaine and levobupivacaine, dosed to produce mild neurotoxicity, administered concurrently with 20% ILE or placebo. Coadministration of ILE decreased the maximum plasma concentration of both ropivacaine and levobupivacaine, with no statistical difference in the dose of LA that produced neurologic symptoms.12 The study is severely limited by its small enrollment, use of proxy outcomes, and lack of clinical difference.
- LAST may progress rapidly from CNS toxicity to cardiotoxicity. Seizures associated with LAST may worsen hypoxia and acidemia. Administration of benzodiazepines to abort seizure-like activity may prevent LA-associated cardiotoxicity and is commonly reported as part of a therapeutic regimen.2, 10, 13
- Sodium bicarbonate administration may overcome sodium channel blockade by LAs and correct acidemia. Evidence to support the use of sodium bicarbonate is limited to case reports as part of a therapeutic regimen10, 13 and 1 porcine RCT demonstrating effective shortening of the QRS interval in bupivacaine toxicity.14
- Bradycardia is the most common cardiovascular sign of LAST.1 Atropine has been used successfully in case reports.15, 16 Several cases reports describe successful utilization of mechanical support such as cardiopulmonary bypass and/or VA-ECMO for patients with LAST and refractory cardiogenic shock.17-21 Unfortunately, lack of widespread availability of VA-ECMO limits the use of these interventions.
Introduction
Acquired methemoglobinemia occurs after an exposure to an oxidant stressor that oxidizes iron in the hemoglobin molecule from the ferrous (Fe2+) state to the ferric (Fe3+) state. In the ferric state, hemoglobin no longer effectively binds and delivers oxygen to end organs. Common sources of oxidant stress that can cause methemoglobinemia include nitrates, nitrites, and many pharmaceuticals (eg, dapsone, benzocaine, phenazopyridine).1-9 Patients with methemoglobinemia can appear cyanotic and dusky and complain of shortness of breath and fatigue. Frequently, a difference is observed between the oxygen saturation measured on pulse oximetry and the oxygen saturation calculated on an arterial blood gas. Severe methemoglobinemia can lead to cardiovascular collapse and death.6, 7, 9
The most widely accepted treatment for methemoglobinemia is methylene blue, which acts as a cofactor to reduce methemoglobin to hemoglobin.10 There are no randomized trials evaluating methylene blue for the treatment for methemoglobinemia, but observational data consistently demonstrate resolution or improvement after methylene blue administration. In addition to methylene blue, other treatment modalities that have been described include exchange transfusion, hyperbaric oxygen therapy, and ascorbic acid.
No studies have examined the treatment of methemoglobinemia in the context of cardiac arrest.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend administering methylene blue for methemoglobinemia. |
| 2b | C-LD | 2. Exchange transfusion may be reasonable as a treatment for methemoglobinemia that is not responsive to methylene blue. |
| 2b | C-LD | 3. Hyperbaric oxygen therapy may be reasonable as a treatment for methemoglobinemia that is not responsive to methylene blue. |
| 3: No Benefit | B-R | 4. N-acetylcysteine is not recommended as a treatment for methemoglobinemia. |
| 3: No Benefit | C-LD | 5. Ascorbic acid is not recommended as a treatment for methemoglobinemia. |
Recommendation-Specific Supportive Text
- Observational studies and published case reports consistently demonstrate that methylene blue effectively reverses methemoglobinemia.1-5, 11 Methylene blue may not improve methemoglobinemia in patients who have glucose-6-phosphate dehydrogenase deficiency.12-14
- Exchange transfusion has been used successfully to treat methemoglobinemia and may be appropriate in patients for whom methylene blue is ineffective.15-20
- Hyperbaric oxygen therapy has been used as monotherapy and in conjunction with other therapies. However, reduction of methemoglobinemia concentrations can be delayed up to several hours.21-23 Its use may be impractical in the setting of cardiopulmonary collapse or cardiac arrest.
- N-acetylcysteine did not reduce sodium-nitrite–induced methemoglobinemia in a double-blind crossover human volunteer study.24
- Ascorbic acid, or vitamin C, has been used to treat methemoglobinemia.17, 25, 26 However, most published case reports demonstrate its use in conjunction with other treatment modalities. The effect is slow and often requires multiple doses over several hours to have any significant effect.6, 26-28 Ascorbic acid is not likely to be effective in resuscitation situations.
Introduction
Since the publication of the last AHA guidelines for the treatment of opioid overdose in 2020,1, 2 the epidemic of opioid poisoning continues to worsen in the United States and in many other nations worldwide. Data from the US National Center for Health Statistics report a staggering 75,673 deaths from opioids in the 12-month period ending in April 2021, a nearly 35% increase from the year before.3 Most deaths are unintentional. Effective primary prevention, emergency treatment, and secondary prevention strategies are urgently needed to address this rapidly escalating crisis.
In formulating these recommendations, the writing group reviewed the 2020 adult, pediatric, and resuscitation education science guidelines,1, 2, 4 the AHA’s 2021 scientific statement on opioid-associated out-of-hospital cardiac arrest,5 and additional literature published since 2019. After careful review, the writing group reaffirms the “2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care,” with additional supporting references and discussion.
As noted in the previous guidelines, isolated opioid toxicity is associated with CNS and respiratory depression that progresses to respiratory arrest, followed by cardiac arrest. Most opioid-associated deaths involve the coingestion of multiple substances or medical and mental health comorbidities.6,7-9 It can be difficult in the hospital setting, and may be impossible in the out-of-hospital setting, to accurately differentiate opioid-associated resuscitative emergencies from other causes of cardiac and respiratory arrest. Opioid-associated resuscitative emergencies are defined by the presence of cardiac arrest, respiratory arrest, or severe life-threatening instability (such as severe CNS or respiratory depression, hypotension, or cardiac dysrhythmia) that is suspected to be due to opioid toxicity.5 In these situations, the mainstay of care remains early recognition and activation of the emergency response system (Figures 1 and 2). Opioid overdoses deteriorate to cardiopulmonary arrest because of loss of airway patency and lack of breathing; therefore, addressing the airway and ventilation in a periarrest patient is of the highest priority.
Naloxone, a µ-opioid receptor antagonist, can restore spontaneous respirations and protective airway reflexes in patients for whom these are impaired due to an opioid overdose. Harmful effects include precipitating opioid withdrawal. Alternatives to naloxone include observation (in patients who are breathing normally regardless of CNS depression) and ventilatory support.
Educating patients with opioid use disorder10, 11 and their friends, families,12 and close contacts10 improves risk awareness, overdose recognition, willingness and ability to administer naloxone, and attitudes toward calling emergency medical services.13, 14 Given the tremendous scope of the problem, widespread community training in cardiopulmonary resuscitation (CPR) and naloxone administration is of growing importance.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. For patients in respiratory arrest, rescue breathing or bag-mask ventilation should be maintained until spontaneous breathing returns, and standard BLS, ALS and/or PALS measures should continue if return of spontaneous breathing does not occur. |
| 1 | C-EO | 2. For patients known or suspected to be in cardiac arrest, in the absence of a proven benefit from the use of naloxone, standard resuscitative measures should take priority over naloxone administration, with a focus on high-quality CPR (compressions plus ventilation). |
| 1 | C-EO | 3. Lay and trained responders should not delay activating emergency response systems while awaiting the patient’s response to naloxone or other interventions. |
| 2a | C-EO | 4. For a patient with suspected opioid overdose who has a definite pulse but no normal breathing or only gasping (ie, a respiratory arrest), in addition to providing standard BLS and/or ALS care, it is reasonable for responders to administer naloxone. |
Recommendation-Specific Supportive Text
- Initial management should focus on support of the patient’s airway and breathing. This begins with opening the airway followed by delivery of rescue breaths, ideally with the use of a bag-mask or barrier device.1, 15, 16 Provision of BLS and ALS care should continue if return of spontaneous breathing does not occur.
- There are no studies demonstrating improvement in patient outcomes from administration of naloxone during cardiac arrest. Provision of CPR should be the focus of initial care.5 Naloxone can be administered along with standard care if it does not delay components of high-quality CPR.
- Early activation of the emergency response system is critical for patients with suspected opioid overdose. Rescuers cannot be certain that the person’s clinical condition is due to opioid-induced respiratory depression alone. This is particularly true in first aid and BLS settings, where determination of the presence of a pulse is unreliable,17, 18 but even trained first responders have difficulty rapidly determining pulselessness.19 Naloxone is ineffective in other medical conditions, including overdose involving nonopioids and cardiac arrest from any cause. Patients who respond to naloxone administration may develop recurrent CNS and/or respiratory depression and require longer periods of observation before safe discharge.20-23
- Twenty-four studies examined the use of naloxone in patients with CNS and/or respiratory depression from opioid poisoning. None compared naloxone administration to standard resuscitation/ventilatory support alone. Seven studies compared intramuscular and intranasal routes of naloxone administration (4 RCT,24-27 3 non-RCT28-30), and 18 other studies31-48 assessed the safety, tolerability, or dosing of naloxone use for opioid poisoning in a variety of settings, mostly out-of-hospital. These studies report that naloxone is safe and effective in treatment of opioid-induced respiratory depression and that major complications are rare and dose related.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | C-LD | 1. After return of spontaneous breathing, patients should be observed in a healthcare setting until the risk of recurrent opioid toxicity is low and the patient’s level of consciousness and vital signs have normalized. |
| 2a | C-LD | 2. If recurrent opioid toxicity develops, repeated small doses or an infusion of naloxone can be beneficial. |
Recommendation-Specific Supportive Text
- Patients who respond to naloxone administration may develop recurrent CNS and/or respiratory depression. Although abbreviated observation periods may be adequate for patients with fentanyl, morphine, or heroin overdose,1, 40, 46, 49-52 longer periods of observation may be required to safely discharge a patient with life-threatening overdose of a long-acting or sustained-release opioid.20-22 Prehospital providers who are faced with the challenge of a patient refusing transport after treatment for a life-threatening overdose are advised to follow local protocols and practices for determination of patient capacity to refuse care.
- Because the duration of action of naloxone may be shorter than the respiratory depressive effect of the opioid, particularly long-acting formulations, repeat doses of naloxone or a naloxone infusion may be required.20-22, 46
| COR | LOE | Recommendations |
|---|---|---|
| 2a | B-R | 1. It is reasonable for lay rescuers to receive training in responding to opioid overdose, including provision of naloxone. |
Recommendation-Specific Supportive Text
- Ten studies assessed the impact of opioid overdose training using a comparator group, with or without randomization, on the ability of persons with opioid use disorder to recognize opioid-associated resuscitation emergencies and/or their willingness to administer naloxone.10-12, 53-59 One study54 found that the rate of naloxone administration was higher in those who had received opioid training compared with those who did not (32% versus 0%), though another study found no difference in the provision of aid between trained and untrained responders.53 Interventions that included skills practice (ie, naloxone administration) were more likely to lead to improved clinical performance compared with interventions without skills practice.12, 60-65
Introduction
Organophosphates (OPs) and carbamates, found in pesticides, nerve agents, and some medications, inhibit acetylcholinesterase, resulting in muscarinic and nicotinic toxicity. They produce parasympathetic excess (bradycardia, bronchospasm, bronchorrhea, miosis, hypersalivation, lacrimation, urination, diarrhea, vomiting, diaphoresis), nicotinic excess (tachycardia, mydriasis, fasciculations progressing to depolarizing neuromuscular blockade and paralysis), and CNS effects (altered mental status, central apnea, seizures).
OPs eventually form a covalent bond with the acetylcholinesterase enzyme, causing permanent inactivation (“aging”). Carbamates spontaneously dissociate from acetylcholinesterase, which is then reactivated.
The cornerstones of treatment include decontamination, atropine, benzodiazepines, and oximes. Dermal decontamination helps prevent further absorption and prevents contamination of caregivers and the care environment. Atropine blocks parasympathetic overstimulation, mitigating bronchorrhea, bradycardia, bronchospasm, and CNS effects. Atropine does not block acetylcholine excess at the neuromuscular junction or nicotinic ganglia and therefore does not reverse paralysis. Benzodiazepines are used to prevent and treat seizures. When administered early (prior to aging), oximes reactivate the acetylcholinesterase enzyme, reversing nicotinic effects to slowly improve respiratory and skeletal muscle strength, though this effect may be agent specific.1-5 Although the available data are not sufficient to support a recommendation for or against oxime use in carbamate poisoning, oximes should not be withheld in cases of cholinesterase poisoning when the class of poison is unknown.
No study to date has specifically evaluated therapy for OP-induced or carbamate-induced cardiac arrest.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | A | 1. Atropine should be given immediately for organophosphate or carbamate poisoning. |
| 1 | B-NR | 2. Early endotracheal intubation should be performed for life-threatening organophosphate or carbamate poisoning. |
| 1 | C-LD | 3. Benzodiazepines should be administered to treat seizures and agitation in the setting of organophosphate or carbamate poisoning. |
| 1 | C-LD | 4. Appropriate personal protective equipment should be used when caring for patients with organophosphate or carbamate exposure. |
| 1 | C-EO | 5. Dermal decontamination should be performed for external organophosphate or carbamate exposure. |
| 2a | A | 6. The use of pralidoxime is reasonable for organophosphate poisoning. |
| 3: No Benefit | C-LD | 7. Use of neuromuscular blockers metabolized by cholinesterase (ie, succinylcholine and mivacurium) are not recommended for patients with organophosphate or carbamate poisoning. |
Recommendation-Specific Supportive Text
- For patients with life-threatening OP or carbamate poisoning, including cardiac arrest, bradycardia, hypotension, bronchorrhea, or bronchospasm, early atropine administration improved survival in a clinical trial.6 Much larger doses of atropine are often required for this indication than for typical bradycardia (Table 2). The initial dose is doubled every 5 minutes until full atropinization is achieved (clear chest on auscultation, heart rate greater than 80/min, systolic blood pressure greater than 80 mmHg). Maintenance of atropinization can be achieved by an atropine infusion.6
- Observational data suggest that patients with significant OP poisoning have better outcomes with early endotracheal intubation.7
- Benzodiazepines such as diazepam (first line) or midazolam have demonstrated efficacy in patients with OP or carbamate-induced seizures and agitation, and effectively manage OP-induced status epilepticus and mitigate neuronal injury in animal models.7-10
- Healthcare providers not wearing appropriate personal protective equipment have developed symptoms consistent with OP exposure after being in close contact with patients poisoned by OPs, including patients with respiratory and dermal exposures only.11-14 Appropriate healthcare provider personal protective equipment depends on the circumstances of the OP exposure and potency of the involved OP.
- Removal of contaminated garments and skin cleansing are highly effective at removing simulated OP exposures.15
- Early administration of oximes, such as pralidoxime, can be considered for significant OP poisoning (especially for those with muscle fasciculations, weakness, or paralysis). Oximes are not universally effective; their effectiveness may be limited by rapid aging of some agents (eg, tabun), their inability to cross the blood–brain barrier, structural differences among OPs, and rapid reinactivation of regenerated acetylcholinesterase in the presence of the poison.5, 16-19
- Neuromuscular blockade from medications metabolized by butyrylcholinesterase (aka pseudocholinesterase), such as succinylcholine, suxamethonium, and mivacurium, can be prolonged by several hours in the context of OP or carbamate poisoning.20-22 Neuromuscular blockers not primarily metabolized by cholinesterases should be used if neuromuscular blockade is needed.
Introduction
Many poisons block cardiac sodium channels with properties similar to Vaughan-Williams class Ia or Ic antidysrhythmics. Sodium channel blocker poisoning causes QRS prolongation, hypotension, seizures, ventricular dysrhythmias, and cardiovascular collapse. Many sodium channel blockers have additional effects on other cardiac receptors and ion channels.1 Although tricyclic and tetracyclic antidepressants (TCAs) are the most commonly described and best-studied sodium channel blockers, many other poisons cause life-threatening sodium channel blockade in overdose (Table 3). Treatment recommendations for poisoning by other sodium channel blockers are often extrapolated from TCA studies. Management of life-threatening poisoning from LAs, whose pharmacologic action is similar to class Ib antidysrhythmics, is discussed in section 9 of this focused update. Treatment of cocaine poisoning, which has toxicity unique from other LAs, is discussed in section 6. Management of chloroquine and hydroxychloroquine poisoning, which is unique but uncommon in North America, is outside the scope of these guidelines.
Characteristic electrocardiogram changes usually precede ventricular dysrhythmias in patients with sodium channel blocker poisoning. These include intraventricular conduction delay (QRS interval prolongation) and the development of a terminal rightward axis deviation, best appreciated in lead aVR (Figure 3).
No studies compare treatments during cardiac arrest from sodium channel blocker poisoning. Human evidence is limited to retrospective observational studies and case reports, in which patients received multiple interventions. The vast majority of these involve TCA poisoning. The therapeutic intervention with the most evidence is hypertonic sodium bicarbonate. Hypertonic sodium administration and induction of alkalemia are variably beneficial in case reports and animal models.2-4 Class Ib antidysrhythmics (eg, lidocaine or phenytoin) and ILE are proposed to treat cardiotoxicity by class Ia and Ic sodium channel blockers.2-5 Other interventions, including sodium bicarbonate and benzodiazepines for seizures, magnesium for WCT, and high-dose glucagon for hypotension, are not supported well enough to inform a recommendation.
Table 3. Selected Sodium Channel Blockers
Carbamazepine
Chloroquine*
Cocaine†
Diphenhydramine
Flecainide
Hydroxychloroquine*
Lamotrigine
Lacosamide
Propafenone
Quinine
Quinidine
Thioridazine
Taxus spp. (yew)
Topiramate
Tricyclic and tetracyclic antidepressants‡
Venlafaxine
Zonisamide
*Treatment of chloroquine and hydroxychloroquine toxicity is outside the scope of this focused update.
†Management of life-threatening cocaine toxicity is discussed in section 6 of this focused update.
‡Common tricyclic and tetracyclic antidepressants include amitriptyline, amoxapine, clomipramine, desipramine, doxepin, imipramine, maprotiline, nortriptyline, protriptyline, and trimipramine.
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. We recommend using hypertonic sodium bicarbonate to treat life-threatening cardiotoxicity from tricyclic and/or tetracyclic antidepressant poisoning. |
| 2a | C-LD | 2. It is reasonable to use hypertonic sodium bicarbonate to treat life-threatening cardiotoxicity caused by poisoning from sodium channel blockers other than tricyclic and tetracyclic antidepressants. |
| 2a | C-LD | 3. It is reasonable to use extracorporeal life support, such as venoarterial extracorporeal membrane oxygenation (VA-ECMO), to treat refractory cardiogenic shock from sodium channel blocker poisoning. |
| 2b | C-LD | 4. It may be reasonable to use Vaughan-Williams class Ib antidysrhythmics (eg, lidocaine) to treat life-threatening cardiotoxicity from class Ia or Ic sodium channel blockers. |
| 2b | C-LD | 5. It may be reasonable to use intravenous lipid emulsion to treat life-threatening sodium channel blocker poisoning refractory to other treatment modalities. |
Recommendation-Specific Supportive Text
1 and 2. Sodium loading and increasing the serum pH (correction of acidemia and/or inducing alkalemia) are each supported for the treatment of hypotension and dysrhythmia from TCA poisoning.3, 6-9 The combination has an additive effect. Hypertonic sodium bicarbonate administration achieves both physiologic goals, though its mechanism is not fully elucidated.7, 10 This practice is supported by case series in TCA poisoning6, 9 and case reports in poisoning by other sodium channel blockers,7, 11-14 though treatment failures are reported and the use of multiple interventions makes it difficult to attribute benefit to any one therapy. Sodium bicarbonate boluses are titrated to resolution of hypotension and QRS prolongation.4, 7, 10 Whether it is superior to then start a continuous infusion or to monitor the patient and administer additional sodium bicarbonate boluses as needed is unsettled.15 Experts recommend avoiding extremes of hypernatremia (serum sodium not to exceed 150–155 mEq/L) and alkalemia (serum pH not to exceed 7.50–7.55) to avoid iatrogenic harm.3, 7, 8, 16, 17 If necessary, serum sodium can be separately increased by administration of hypertonic saline,18 and pH can be controlled by adjusting minute ventilation in intubated patients.19
- Extracorporeal support, including VA-ECMO, has been used successfully in patients with refractory cardiogenic shock from sodium channel blocker poisoning.20-24 Controlled observational studies and clinical trial data do not exist. Further discussion of the use of VA-ECMO in poisoning is provided in section 15.
- Lidocaine, a class Ib antidysrhythmic, competes with Ia and Ic antidysrhythmics for binding at the sodium channel and dissociates from the receptor more rapidly than Ia or Ic agents, such as TCAs, and therefore does not depress phase 0 depolarization.2 The use of lidocaine to treat WCT from TCA overdose is supported by animal studies and human case reports.2 A similar role for phenytoin, another class Ib antidysrhythmic, is supported by human case reports,2, 25 though not consistently by animal studies.26, 27 Lidocaine or phenytoin are second-line therapies, after hypertonic sodium bicarbonate.
- Most sodium channel blockers are highly lipophilic. Several case reports describe temporal improvement following ILE administration,28-30 including successful treatment of TCA-induced cardiac arrest.31-33 These case reports are subject to publication bias. An RCT, published in abstract form only, found no benefit from ILE administration in treatment of hypotension or electrocardiogram abnormalities from TCA poisoning.34 Furthermore, ILE administration may increase drug absorption in oral overdose,35 and animal studies are not supportive.36 The Lipid Emulsion Workgroup recommends the use of ILE for life-threatening TCA toxicity “if other therapies fail/in last resort,” or after failure of standard therapies, but “not as first-line therapy.”37 The Lipid Emulsion Workgroup makes a neutral recommendation for cardiac arrest.
Introduction
The hallmark of sympathomimetic poisoning is increased activity of the adrenergic nervous system. Amphetamines, cathinones, and some synthetic cannabinoid receptor agonists produce sympathomimetic poisoning. When treatment is required, clinicians are rarely able to determine which specific substance was used, and treatment must be based on presenting signs and symptoms and limited available history. Management of severe cocaine poisoning is discussed separately in section 6. Complications of sympathomimetic poisoning result from excessive catecholamine release and attendant increase in metabolic and psychomotor activity. Patients present on a spectrum of severity. Clinical manifestations include tachycardia, hypertension, agitation, seizures, hyperthermia, rhabdomyolysis, and acidosis.1-4
Sympathomimetic poisoning can cause sudden cardiac arrest, presenting as ventricular fibrillation, /ventricular tachycardia, or pulseless electrical activity..5-8 Vasospasm can cause myocardial infarction, even in patients with normal coronary arteries.5, 9-11 A transient stress cardiomyopathy is also reported in sympathomimetic-poisoned patients; this condition can be fatal, but it resolves spontaneously in survivors.10, 12-15 Hyperthermia is a severe and rapidly life-threatening clinical manifestation.2, 3, 6, 16, 17 Physical restraints may be temporarily necessary, but their prolonged use may exacerbate hyperthermia and agitation.
Although many clinical trials and observational studies have been published comparing various sedatives for patients with severe psychomotor agitation, none have focused on the prevention or treatment of cardiac arrest. Therefore, evidentiary support for management is primarily from nonhuman experiments, published cases, and expert opinion. Although there is no direct antidote to sympathomimetic poisoning, sedatives treat psychomotor agitation that results in delirium, rhabdomyolysis, and hyperthermia.18-23 In some cases, large doses of sedatives are required.4, 24, 25 External cooling directly treats hyperthermia, potentially reducing brain and other organ injury.6
| COR | LOE | Recommendations |
|---|---|---|
| 1 | B-NR | 1. Sedation is recommended for severe agitation from sympathomimetic poisoning. |
| 1 | C-LD | 2. Rapid external cooling is recommended for life-threatening hyperthermia from sympathomimetic poisoning. |
| 2a | C-EO | 3. Vasodilators, such as phentolamine and/or nitrates, are reasonable for coronary vasospasm from sympathomimetic poisoning. |
| 2a | C-EO | 4. Mechanical circulatory support, such as intra-aortic balloon pump or veno-arterial extracorporeal membrane oxygenation (VA-ECMO, is reasonable for cardiogenic shock from sympathomimetic poisoning refractory to other treatment measures. |
| 3: Harm | C-LD | 5. Prolonged use of physical restraint without sedation is potentially harmful. |
Recommendation-Specific Supportive Text
- Sedatives (eg, benzodiazepines, antipsychotics, ketamine) have been used in nonhuman experiments and case reports to treat sympathomimetic poisoning.26, 27 Sedatives treat delirium and control psychomotor agitation that produces heat and rhabdomyolysis. Antipsychotics control agitation. Benzodiazepines control agitation, relax muscles, and treat seizures. Although several clinical trials compare specific agents for severe psychomotor agitation, it is difficult to separate patients with sympathomimetic poisoning from other patients in these studies, and cardiac arrest was rare.28-31
- Hyperthermia is rapidly life-threatening in sympathomimetic poisoning.23 External cooling has been used to treat hyperthermia in patients with sympathomimetic poisoning.6, 32-34 Evaporative or immersive cooling modalities reduce temperature more rapidly than cooling blankets or the application of cold packs.35, 36
- Vasodilators, including nitrates and α-adrenergic receptor antagonists, have been used to treat coronary vasospasm, reversing electrocardiographic and biochemical markers of ischemia in sympathomimetic-poisoned patients.21, 37, 38
- Mechanical circulatory support, including VA-ECMO14, 39 and intra-aortic balloon pump,10, 40 has been used successfully to support cardiac output in patients in cardiogenic shock while stress cardiomyopathy resolves. Stress (takotsubo) cardiomyopathy can be fatal, but it often spontaneously resolves in days to weeks with circulatory support.
- Although physical restraints may be temporarily necessary, their sustained use without effective sedation is associated with death in patients with severe agitation.38, 41-43 Restraints should be removed as soon as safely possible.
Introduction
Venoarterial extracorporeal membrane oxygenation (VA-ECMO) is a resuscitative measure providing both cardiac and pulmonary support.1 In the setting of poisoning, VA-ECMO treats refractory cardiogenic shock by providing mechanical circulatory support while the offending poison is eliminated. The use of VA-ECMO for poisoning is increasing.2 There are no RCTs comparing the use of VA-ECMO versus supportive care for the poisoned patient. An RCT comparing VA-ECMO to standard care for patients with refractory out-of-hospital cardiac arrest found improved survival with VA-ECMO.3 However, this study excluded patients with drug overdose. Observational studies demonstrate that patients with cardiac arrest or refractory shock due to poisoning who are managed with VA-ECMO have lower mortality than other patients treated with VA-ECMO as well as lower mortality compared with poisoned patients treated with standard critical care and antidotal therapy alone.4
The use of VA-ECMO in the poisoned patient is limited by availability, logistics of transport, patient comorbidities, and risks inherent to the procedure. The pathophysiology of the specific poisoning and the clinical features of the patient must both be considered in the decision to initiate VA-ECMO. In particular, VA-ECMO does not generally correct distributive shock or reverse cellular injury. A multidisciplinary approach, including consultation from a poison center or medical toxicologist, is helpful to determine the appropriateness of VA-ECMO in specific cases.
The use of VA-ECMO in the context of cardiac arrest is also termed extracorporeal cardiopulmonary resuscitation (ECPR). Current AHA guidelines for ALS resuscitation state that “ECPR may be considered for select cardiac arrest patients for whom the suspected cause of the cardiac arrest is potentially reversible during a limited period of mechanical circulatory support (COR 2a, LOE C-LD).”5 The most recent PALS guidelines state, “ECPR may be considered for pediatric patients with cardiac diagnoses who have IHCA in settings with existing ECMO protocols, expertise, and equipment (COR 2b, LOE C-LD).”6
Other forms of mechanical circulatory support, such as implanted left ventricular assist devices and percutaneous mechanical circulatory support devices (intra-aortic balloon pump and newer devices) each has its own risks and benefits and may be considered for clinical scenarios similar to those described here.
| COR | LOE | Recommendations |
|---|---|---|
| 2a | C-LD | 1. It is reasonable to use VA-ECMO for persistent cardiogenic shock or cardiac arrest due to poisoning that is not responsive to maximal treatment measures. |
| 2a | C-LD | 2. It is reasonable to use VA-ECMO for persistent dysrhythmias due to poisoning when other treatment measures fail. |
| 2b | C-EO | 3. The effectiveness of VA-ECMO for poisoned patients with cardiovascular collapse from causes other than cardiogenic shock has not been established. |
- In 1 retrospective review of 64 patients treated with VA-ECMO for cardiac arrest or refractory shock regardless of cause, cardiotoxic poisoning was independently associated with survival.7 In an observational study of 62 patients with cardiac arrest or severe shock following poisoning, VA-ECMO was associated with reduced mortality compared with standard care alone.4 There are risks for significant complications, including limb ischemia, bleeding, stroke, and infection.1, 8
- For patients with persistent non-perfusing dysrhythmias, VA-ECMO provides forward blood flow to allow for poison elimination. Case reports describe the use of VA-ECMO to support poisoned patients with persistent dysrhythmias.9-14 This literature is limited by publication bias.
- In one case series, patients with hematologic and metabolic poisons had higher mortality on VA-ECMO compared with other poisonings.2 The efficacy of VA-ECMO is undefined in poisonings that cause refractory vasodilatory shock with preserved cardiac function, direct cellular toxicity, disruption of cellular oxygen use, or poisonings that are universally fatal despite temporary cardiac support.
As part of the overall work for development of these guidelines, the writing group was able to review a large amount of literature concerning the management of adult cardiac arrest. One expected challenge faced through this process was the lack of data in many areas of cardiac arrest research. This challenge was faced in both the 2010 Guidelines and 2015 Guidelines Update processes, where only a small percent of guideline recommendations (1%) were based on high-grade LOE (A) and nearly three quarters were based on low-grade LOE (C).1
Similar challenges were faced in the 2020 Guidelines process, where a number of critical knowledge gaps were identified in adult cardiac arrest management. These topics were identified as not only areas where no information was identified but also where the results of ongoing research could impact the recommendation directly. Throughout the recommendation-specific text, the need for specific research is identified to facilitate the next steps in the evolution of these questions. Critical knowledge gaps are summarized in Table 4.
Table 4. 2020 Adult Guidelines Critical Knowledge Gaps
| Sequence of Resuscitation |
|
|---|---|
| Initiation of resuscitation | What are optimal strategies to enhance lay rescuer performance of CPR? |
| Metrics for high-quality CPR | What is optimal for the CPR duty cycle (the proportion of time spent in compression relative to the total time of the compression-plus-decompression cycle)? |
| Metrics for high-quality CPR | What is the validity and reliability of ETCO2 in nonintubated patients? |
| Metrics for high-quality CPR | For patients with an arterial line in place, does targeting CPR to a particular blood pressure improve outcomes? |
| Metrics for high-quality CPR | How does integrated team performance, as opposed to performance on individual resuscitation skills, affect resuscitation outcomes? |
| Defibrillation | Is there an ideal time in the CPR cycle for defibrillator charging? |
| Defibrillation | Can artifact-filtering algorithms for analysis of ECG rhythms during CPR in a real-time clinical setting decrease pauses in chest compressions and improve outcomes? |
| Defibrillation | Does preshock waveform analysis lead to improved outcome? |
| Defibrillation | Do double sequential defibrillation and/or alternative defibrillator pad positioning affect outcome in cardiac arrest with shockable rhythm? |
| Vascular access | Is the IO route of drug administration safe and efficacious in cardiac arrest, and does efficacy vary by IO site? |
| Vasopressor medications during cardiac arrest | Does epinephrine, when administered early after cardiac arrest, improve survival with favorable neurological outcome? |
| Nonvasopressor medications during cardiac arrest | Do antiarrhythmic drugs, when given in combination for cardiac arrest, improve outcomes from cardiac arrest with shockable rhythm? |
| Nonvasopressor medications during cardiac arrest | Do prophylactic antiarrhythmic medications on ROSC after successful defibrillation decrease arrhythmia recurrence and improve outcome? |
| Nonvasopressor medications during cardiac arrest | Do steroids improve shock or other outcomes in patients who remain hypotensive after ROSC? |
| Adjuncts to CPR | Does the use of point-of-care cardiac ultrasound during cardiac arrest improve outcomes? |
| Adjuncts to CPR | Is targeting a specific ETCO2 value during CPR beneficial, and what degree of rise in ETCO2 indicates ROSC? |
| Termination of resuscitation | Can ETCO2 be used for intra-arrest prognostication, in combination with other metrics? |
| Termination of resuscitation | Can point-of-care cardiac ultrasound, in conjunction with other factors, inform termination of resuscitation? |
| Advanced Techniques and Devices for Resuscitation |
|
| Advanced airway placement | What is the optimal approach to advanced airway management for IHCA? |
| Advanced airway placement | There is a need for further research specifically on the interface between patient factors and the experience, training, tools, and skills of the provider when choosing an approach to airway management. |
| Advanced airway placement | What is the specific type, amount, and interval between airway management training experiences to maintain proficiency? |
| Alternative CPR techniques and devices | Which populations are most likely to benefit from ECPR? |
| Specific Arrhythmia Management |
|
| Atrial fibrillation or flutter with rapid ventricular response | What is the optimal energy needed for cardioversion of atrial fibrillation and atrial flutter? |
| Bradycardia | What is the optimal approach, vasopressor or transcutaneous pacing, in managing symptomatic bradycardia? |
| Care After ROSC | |
| Postresuscitation care | Does avoidance of hyperoxia in the postarrest period lead to improved outcomes? |
| Postresuscitation care | What is the effect of hypocarbia or hypercarbia on outcome after cardiac arrest? |
| Postresuscitation care | Does the treatment of nonconvulsive seizures, common in postarrest patients, improve patient outcomes? |
| Postresuscitation care | What are the optimal pharmacological treatment regimens for the management of postarrest seizures? |
| Postresuscitation care | Do neuroprotective agents improve favorable neurological outcome after arrest? |
| Postresuscitation care | What is the most efficacious management approach for postarrest cardiogenic shock, including pharmacological, catheter intervention, or implantable device? |
| Postresuscitation care | Is there a role for prophylactic antiarrhythmics after ROSC? |
| Targeted temperature management | Does targeted temperature management, compared to strict normothermia, improve outcomes? |
| Targeted temperature management | What is the optimal temperature goal for targeted temperature management? |
| Targeted temperature management | What is the optimal duration for targeted temperature management before rewarming? |
| Targeted temperature management | What is the best approach to rewarming postarrest patients after treatment with targeted temperature management? |
| PCI after cardiac arrest | Does emergent PCI for patients with ROSC after VF/VT cardiac arrest and no STEMI but with signs of shock or electric instability improve outcomes? |
| Neuroprognostication | What is the interrater agreement for physical examination findings such as pupillary light reflex, corneal reflex, and myoclonus/status myoclonus? |
| Neuroprognostication | Can we identify consistent NSE and S100B thresholds for predicting poor neurological outcome after cardiac arrest? |
| Neuroprognostication | Are NSE and S100B helpful when checked later than 72 h after ROSC? |
| Neuroprognostication | Are glial fibrillary acidic protein, serum tau protein, and neurofilament light chain valuable for neuroprognostication? |
| Neuroprognostication | More uniform definitions for status epilepticus, malignant EEG patterns, and other EEG patterns are needed to be able to compare prognostic values across studies. |
| Neuroprognostication | What is the optimal timing for head CT for prognostication? |
| Neuroprognostication | Is there a consistent threshold value for prognostication for GWR or ADC? |
| Neuroprognostication | Standardization of methods for quantifying GWR and ADC would be useful. |
| Recovery |
|
| Recovery and survivorship after cardiac arrest | What do survivor-derived outcome measures of the impact of cardiac arrest survival look like, and how do they differ from current generic or clinician-derived measures? |
| Recovery and survivorship after cardiac arrest | Are there in-hospital interventions that can reduce or prevent physical impairment after cardiac arrest? |
| Recovery and survivorship after cardiac arrest | Which patients develop affective/psychological disorders of well-being after cardiac arrest, and are they treatable/preventable/recoverable? |
| Recovery and survivorship after cardiac arrest | Does hospital-based protocolized discharge planning for cardiac arrest survivors improve access to/ referral to rehabilitation services or patient outcomes? |
| Special Circumstances of Resuscitation |
|
| Accidental hypothermia | What combination of features can identify patients with no chance of survival, even if rewarmed? |
| Accidental hypothermia | Should severely hypothermic patients receive intubation and mechanical ventilation or simply warm humidified oxygen? |
| Accidental hypothermia | Should severely hypothermic patients in VF who fail an initial defibrillation attempt receive additional defibrillation? |
| Accidental hypothermia | Should severely hypothermic patients in cardiac arrest receive epinephrine or other resuscitation medications? If so, what dose and schedule should be used? |
| Drowning | In what situations is attempted resuscitation of the drowning victim futile? |
| Drowning | How long after mild drowning events should patients be observed for late-onset respiratory effects? |
| Electrolyte abnormalities | What is the optimal treatment for hyperkalemia with life-threatening arrhythmia or cardiac arrest? |
| Opioid overdose | What is the minimum safe observation period after reversal of respiratory depression from opioid overdose with naloxone? Does this vary based on the opioid involved? |
| Opioid overdose | Is there benefit to naloxone administration in patients with opioid-associated cardiac arrest who are receiving CPR with ventilation? |
| Opioid overdose | What is the ideal initial dose of naloxone in a setting where fentanyl and fentanyl analogues are responsible for a large proportion of opioid overdose? |
| Opioid overdose | In cases of suspected opioid overdose managed by a non–healthcare provider who is not capable of reliably checking a pulse, is initiation of CPR beneficial? |
| Pregnancy | What is the ideal timing of PMCD for a pregnant woman in cardiac arrest? |
| Pulmonary embolism | Which patients with cardiac arrest due to “suspected” pulmonary embolism benefit from emergency thrombolysis during resuscitation? |
| Toxicity: β-adrenergic blockers and calcium channel blockers | What is the ideal sequencing of modalities (traditional vasopressors, calcium, glucagon, high-dose insulin) for refractory shock due to β-adrenergic blocker or calcium channel blocker overdose? |
| Toxicity: local anesthetics | What are the ideal dose and formulation of IV lipid emulsion therapy? |
| Toxicity: carbon monoxide, digoxin, and cyanide | Which patients with cyanide poisoning benefit from antidotal therapy? |
| Toxicity: carbon monoxide, digoxin, and cyanide | Does sodium thiosulfate provide additional benefit to patients with cyanide poisoning who are treated with hydroxocobalamin? |
ADC indicates apparent diffusion coefficient; CPR, cardiopulmonary resuscitation; CT, computed tomography; ECG, electrocardiogram; ECPR, extracorporeal cardiopulmonary resuscitation; EEG, electroencephalogram; ETCO2, end-tidal carbon dioxide; GWR, gray-white ratio; IHCA, in-hospital cardiac arrest; IO, intraosseous; IV, intravenous; NSE, neuron-specific enolase; PCI, percutaneous coronary intervention; PMCD, perimortem cesarean delivery; ROSC, return of spontaneous circulation; S100B, S100 calcium binding protein; STEMI, ST-segment elevation myocardial infarction; and VF, ventricular fibrillation.
The American Heart Association requests that this document be cited as follows: Panchal AR, Bartos JA, Cabañas JG, Donnino MW, Drennan IR, Hirsch KG, Kudenchuk PJ, Kurz MC, Lavonas EJ, Morley PT, O’Neil BJ, Peberdy MA, Rittenberger JC, Rodriguez AJ, Sawyer KN, Berg KM; on behalf of the Adult Basic and Advanced Life Support Writing Group. Part 3: adult basic and advanced life support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2020;142(suppl 2):S366–S468. doi: 10.1161/CIR.0000000000000916
- Ashish R. Panchal, MD, PhD, Chair
- Jason A. Bartos, MD, PhD
- José G. Cabañas, MD, MPH
- Michael W. Donnino, MD
- Ian R. Drennan, ACP, PhD(C)
- Karen G. Hirsch, MD
- Peter J. Kudenchuk, MD
- Michael C. Kurz, MD, MS
- Eric J. Lavonas, MD, MS
- Peter T. Morley, MBBS
- Brian J. O’Neil, MD
- Mary Ann Peberdy, MD
- Jon C. Rittenberger, MD, MS
- Amber J. Rodriguez, PhD
- Kelly N. Sawyer, MD, MS
- Katherine M. Berg, MD, Vice Chair
On behalf of the Adult Basic and Advanced Life Support Writing Group
The writing group acknowledges the following contributors: Julie Arafeh, RN, MSN; Justin L. Benoit, MD, MS; Maureen Chase; MD, MPH; Antonio Fernandez; Edison Ferreira de Paiva, MD, PhD; Bryan L. Fischberg, NRP; Gustavo E. Flores, MD, EMT-P; Peter Fromm, MPH, RN; Raul Gazmuri, MD, PhD; Blayke Courtney Gibson, MD; Theresa Hoadley, MD, PhD; Cindy H. Hsu, MD, PhD; Mahmoud Issa, MD; Adam Kessler, DO; Mark S. Link, MD; David J. Magid, MD, MPH; Keith Marrill, MD; Tonia Nicholson, MBBS; Joseph P. Ornato, MD; Garrett Pacheco, MD; Michael Parr, MB; Rahul Pawar, MBBS, MD; James Jaxton, MD; Sarah M. Perman, MD, MSCE; James Pribble, MD; Derek Robinett, MD; Daniel Rolston, MD; Comilla Sasson, MD, PhD; Sree Veena Satyapriya, MD; Travis Sharkey, MD, PhD; Jasmeet Soar, MA, MB, BChir; Deb Torman, MBA, MEd, AT, ATC, EMT-P; Benjamin Von Schweinitz; Anezi Uzendu, MD; and Carolyn M. Zelop, MD.
The writing group would also like to acknowledge the outstanding contributions of David J. Magid, MD, MPH.
Open table in a new window.
Open table in a new window.
 Login
Login